Review Article | DOI: https://doi.org/10.31579/2834-8508/017
Threat of Prostate Cancer by Diabetes: The Combination effects of Metformin and Sulfonylurea
1Department of Psychology, Foundation University Islamabad, Pakistan
2Department of Anatomy, University of Health Sciences Lahore, Pakistan
*Corresponding Author: Ammarah Saher, Department of Psychology, Foundation University Islamabad, Pakistan.
Citation: Ammarah Saher, Rabia Latif (2023), Threat of Prostate Cancer by Diabetes: The Combination effects of Metformin and Sulfonylurea. Archives of Clinical and Experimental Pathology. 2(5); Doi:10.31579/2834-8508/017
Copyright: © 2023 Ammarah Saher, This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 18 August 2023 | Accepted: 08 September 2023 | Published: 25 September 2023
Keywords: prostate cancer, diabetes, metformin, sulfonylurea, anticancer effects
Abstract
Cancer and diabetes are chronic, multifarious, and potentially lethal diseases. Diabetes is the seventh leading cause of death, while cancer is the second leading cause of death, the latter being possibly also under reported. There is an increasing body of data published in current years, which indicates a significant increase in the incidence of cancer in diabetic patients. The predicted worldwide incidence of diabetes grew from 171 million in 2000 to 366 million in 2030. For all people over 55, about 16.7% have diabetes and 50% have cancer. Overall, diabetes is present in 8–18% of cancer patients. The medicine of selection used for the cure of type 2 diabetes is metformin & sulfonylurea. The proof obtained from basically of clinical, population, and science-based studies support metformin along with sulfonylurea as anticancer influence. Nevertheless, contingent controlled clinical testing does not offer ample evidence that metformin, combined with sulfonylurea, has a significant protective effect on cancer death or incidence. Some of the most advantages drawbacks of these tastings are the short follow-up time. In order to present the best response to this challenge, more long-term contingent maintained clinical testing specifically designed to evaluate metformin along with the effect of sulfonylurea on cancer risk is required.
1. Introduction
Diabetes Mellitus is a significant as well as increasing health issue worldwide and is connected with extreme chronic and acute complications that adversely affect mutually the survival of the individuals affected and the quality of life. Cancer can be preferred in diabetic patients by: i) Common mechanisms which enhance the initiation as well as progression of cancer in every organ due to alterations (i.e. hyperinsulinemia or hyperglycemia or drugs) affecting every tissue; and ii) Site-specific mechanisms that cause the cancer of a given organ. Diabetes mellitus has been linked with an elevated risk of many cancers, including liver, pancreatic cancer, endometrium, colon, biliary tract, oesophagus, and thyroid. Researchers studying the relationship between prostate cancer and diabetes have shown, however, that diabetes can be associated with a reduced risk of developing this cancer [21]. A meta-analysis of studies reviewed published in 2002 was undertaken by Bonovas et al. and was published in 2004 [22]. The study found whether diabetic patients had a statistically substantial (9%) reduction in the probability of contracting prostate carcinoma. These were stated that there had been virtually no discrepancy among prospective and particular instance trials, nor did any particular analysis substantially alter the cumulative absolute risk. Although Bonovas et al. carried out their meta-analysis, six related research examining the relationship among prostate cancer and diabetes mellitus have been published. These findings are both predictive and observational research and contain f11,000 new cases of prostate cancer. Mostly with additional statistical strength of providing >20,000 incidents of prostate cancer relative to f9,000 cases, in order to gain insight into the pathophysiology of prostate cancer, we plan to further examine this relationship by performing a revised comprehensive meta-analysis. Medically diagnosed prostate cancer is also a recurrent cancer with a high risk for metastases. Prostate cancer screening is capable of identifying disease at a much younger and/or localised level, and it is not yet known if either of these PSA cancers has the ability to progress into pathological changes diseases.
2 Mostly with additional research performed in the PSA test period, we are now in a position to do a subgroup study of the males diagnosed before and after this point. This subgroup study would help us to evaluate the impact that testing can function in the documented relationship between prostate cancer and diabetes mellitus. Given the limitations of the source data, we have not been able to precisely examine the distinctions between advanced and localized tumors, and the prePSA and PSA screening comparisons may offer insight into several different forms of prostate cancer. In regard to PSA-testing subgroups and subgroups close to those mentioned by Bonovas et al., in this metaanalysis, we are able to examine those that did or did not monitor body mass index, including or omitted prostate cancer cases that were detected over the first year after the detection of diabetes mellitus, and trials that accounted for less than three or three or more covariates. Both metformin and sulfonylureas have long been used in the treatment of non-insulin-dependent diabetes mellitus in Europe, but now these are available in the United States. Sulfonylureas suppress hyperglycemia by stimulating insulin secretion [23] and can also encourage overweight, hypoglycemia, and hyperinsulinemia. Metformin tends to decrease insulin resistance [24], does not encourage hypoglycemia, and may minimize body weight. Its disadvantages are frequent gastrointestinal side effects and, exceptionally, lactic acidosis. However, this problem can indeed be avoided by prudently respecting contraindications [25]. Non-insulin-dependent diabetes mellitus typically occurs in insulin-resistant respondents that are unable to completely compensate for increased insulin secretion due to decreased j8-cell function [26]. Therefore, combined therapy with sulfonylurea and metformin would tend to be reasonable. There are, however, few reports of how this hybrid treatment can be optimized. Popular therapeutic practice in Sweden and several other countries is the inclusion of metformin only when sulfonylurea treatment fails, a practice that can have some effect but seldom permits the normalization of hyperglycemia [27]. A meta-analysis of 10 longitudinal studies identified a substantial cumulative, modified risk ratio for all-cancer incidence (RR = 0.89, 93 % CI, and 0.83 to 0.96). There was an elevated risk for both women (RR = 0.97, CI, and 0.87 to 1.24) and men (RR = 0.93, CI, and 0.85 to 1.19) [3]. A recent meta-analysis [2] involving 14 studies performed in the pre-PSA period (i.e.) widespread use of prostate-specific antigen screening in prostate cancer; [4] and 3 additional studies conducted in the PSA period found a substantially lesser risk of diabetic patients. Deng et al. registered a rise in colorectal cancer incidence of 26 %. The rate was comparable for both males and females. These findings were obtained without heterogeneity between the studies from 6 casecontrol and 14 cohort studies (P = 0.196) [1]. There are contradictory data available about the possibility of prostate cancer in type 2 diabetes. A current meta-analysis of 24 testing and 13 case-control studies investigated the relationship among the type prostate cancer risk and 2 diabetes and demonstrated a strong inverse association (RR 1.06, 85 % CI: 1.02– 1.12). In conclusion, due to specific study plan, incomplete classification of DM, failure to regard as possible confounders such as length of diabetes, obesity, and treatment, as well as variable specified maintain population; the epidemiological studies cited over may be incomplete biased by related heterogeneities. Nonetheless, the overall increased risk of multiple forms of cancer arising in diabetic patients must be measured well known. With diabetes, the prevalence of pancreas, breast, liver, urinary tract, colorectal, and female reproductive organ cancer rises marginally too significantly, as well as a minor decrease in the threat of prostate cancer.
2. Effects of Metformin & Sulfonylurea and Cancer
A. Historical Perspective of Metformin & Sulfonylurea
Biguanides were identified as Galega officinalis' active ingredient in the 1920s, and were established as therapeutics in the 1950s. Metformin, buformin, and phenformin have all been established and used to treat type 2 diabetes [6]. The issue with the latter two, namely buformin and phenformin, was toxicity associated with lactic acidosis. Therefore, by the 1970s, 3 they were removed from the market. Nevertheless, metformin demonstrated to be safe and has been accepted as the most successful and the safest treatments for type 2 diabetes care. After many years, it started "a new life for an old drug." Biguanides' role in metabolic rehabilitation and metabolic immunotherapy has released a new window into the future: metformin use beyond diabetes [5]. In 1942, Janbon et al. were discovered Sulfonylureas, experiential that hypoglycemia in experimental animals was caused by certain sulfonamides. Carbutamide (1- butyl-3-sulfonylurea) was synthesised from this observation. Carbutamide was the most common sulfonylurea to be used to cure diabetes, but was later removed from the market due to its unfavorable effects on the bone marrow [7]. Several sulfonylureas became available through the 1960s; they are generally divided into two generations. Glipizide, gliclazide, glimepiride, and glibenclamide are commonly used as secondgeneration sulfonylureas, although drugs of the first generation are chlorpropamide and tolbutamide are no longer used.
B. Mechanism of Metformin Action
Adenosine monophosphate kinase (AMPK) is the sensor of the cellular energy within the cytoplasm. It acts to control metabolism within cells. They are:
1. Activated by cellular stress
2. An attractive target for anticancer therapy
3. Cellular energy sensor
4. Inadequate activity allows unrestrained cell growth
5. Interaction between metabolism and cancer
The tumor suppressor gene produces a protein called the liver kinase B1 (LKB1). AMPK catalytic subunit phosphorylation happens within the sight of LKB1, and is encouraged by AMP. Rising intracellular degrees of adenosine monophosphate (AMP) actuate AMPK [10]. Initiation of AMPK adds to the restraint of the mammalian objective of rapamycin (mTOR) flagging, bringing about deregulation of liver gluconeogenesis.
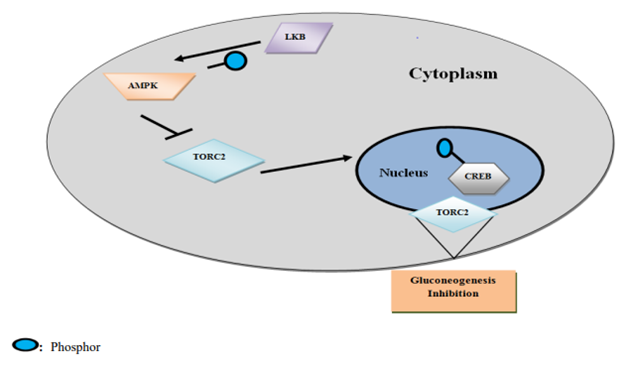
Figure 1. Assimilation of Energy Sensing Pathways through the TORC2 Coactivator
AMPK's role in energy balance regulation is demonstrated in the Table 1.
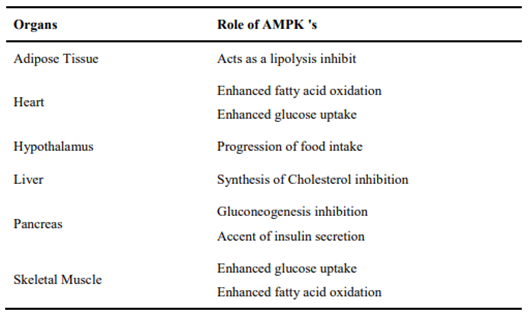
Table 1. AMPK 's Role in Energy Balance Regulation
On the cellular level, metformin exerts both direct and indirect actions. Its direct effect is arbitrated by reduction of the mTOR signaling pathway and AMPK activation that causes the cell proliferation in cancer cells, protein synthesis, and gluconeogenesis inhibition in the liver [8]. The indirect effects of metformin are controlled by its ability to decrease the level of circulating insulin and reduce blood glucose. AMPK is activated by metformin in the skeletal muscles and liver. This decreases gluconeogenesis in the liver and facilitates the absorption of glucose by the peripheral tissues, leading to lesser insulin content and blood glucose. Insulin has been recognized to contain mitogenic activity and may potentially contain a calming consequence on tumor cell growth [9]. While various anticancer effects have been identified for metformin, suppression of the LKB1 medicated mTOR signaling appears to be the basic mechanism of metformin's anti-cancer activity. Table 2 shows the potential impact of metformin on cancer.

Table 2. Potential Impact of Metformin on Cancer
C. Mechanism of Sulfonylureas Action
The important function of sulfonylureas is to increase concentrations of plasma insulin; thus they are only successful when there are residual β-cells of the pancreas. The increase in plasma insulin concentrations presents for two reasons. Firstly, pancreas β-cells promote insulin secretion and secondly, there is a drop in insulin hepatic clearance. This second effect, particularly occurs after there has been an increase in insulin secretion. In addition, the insulin and insulin response levels for glucose increase rapidly in the first month of treatment, leading to lower blood glucose. During this time, the baseline and the activated insulin levels are reduced relative to those considered at the start of therapy; however, blood glucose values remains constant. The explanation for that comment is not properly mentioned. The mechanism is now understood about the secretive behavior of sulfonylureas. They act by binding to the unique sulfonylurea receptor on β-pancreatic cells, blocking the inflow of potassium (K+) during the ATP-dependent channel: the flow of K+ inside the β-cell goes to zero, the cell membrane is depolarized, thus eliminating the electrical screen that prevents the transmission of calcium into the cytosol. The improved inflow of calcium into β-cells triggers the contraction of the actomyosin filaments responsible for insulin exocytosis, and is therefore promptly secreted in large quantities as seen in Figure 2.
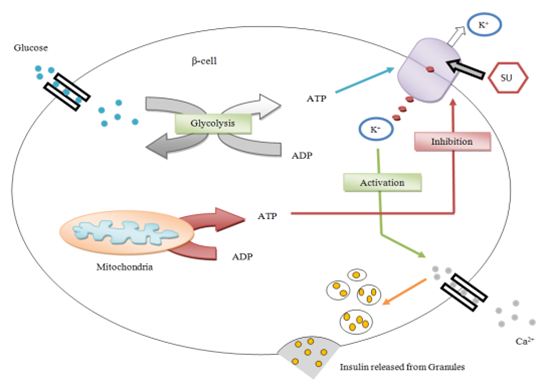
Figure 2. Mechanism of Sulfonylureas Action
In particular, glibenclamide has a high affinity to the SUR1, a protein of 1581 amino acids. SUR1 is a member of the superfamily ABC with two NBF-2 and NBF-1. Every nucleotide binding fold has the Walker A as well as B motifs and the SGGQ ABC signature, and the functional activities of ABC proteins are important in the regulation of nucleotides. SUR1 has three TMD, such as TMD0, TMD1 and TMD2, consisting of increasingly numerated segments of 5, 6 and 6 TM respectively. TMD0 comprises segments of TM from 0 to 4, TMD1 include segments of TM from 5 to 10 and TMD2 comprises segments of TM from 11 to 16. Among pancreatic islets SUR1 is articulated at elevated levels. SUR1 is present in the cortex, too. There is a second type of receptor for sulfonylureas; it is named SUR2 or SUR2A, and it is an isoform of SUR1. SUR2A is a protein of 1456 amino acids sharing an amino acid association of 68% with SUR1.
Glibenclamide has a weak affinity to SUR2A. Various variants of SUR2A were also described. One of them, SUR2B, diverges from SUR2A in the C-terminus by 42 amino acids; anywhere it is identical to SUR1, instead. Though SUR2A is mainly articulated in the skeletal muscle and heart, SUR2B is generally articulated in other tissues. A two-site model such as A and B site for the contact between glinides, sulfonylureas, and SUR had been proposed in the past. The A site is located on the eighth cytosolic loop, which is unique to SUR1 between segments 15 and 16 of TM. Then the B site consists of the third cytosolic loop between TM segments 6 and 7, which is extremely common in all SURs. Glinides and sulfonylureas can be classified into three classes according to these different contact sites. The first of these includes gliclazide, tolbutamide as well as nateglinide, which are molecules that specifically attaches the SUR1 A site, as the second group, which contains glibenclamide as well as glimepiride, binds non-specifically the SUR1 and SUR2A B sites and the SUR1 A site, and eventually, the third group such as repaglinide and meglitinide, which attaches to the SUR1 and SUR2A B sites. In addition to the "first step," sulfonylureas as well as improve the "second phase" of insulin secretion, which starts 10 min later as insulin granules are translated to the β-cell membrane. This second step includes the gradual development of fresh insulin granules, which is simply potential if the future β-cell is conserved. It is very significant to note that sulfonylurea-induced insulin discharge is independent of glucose levels, and this may raise the hypoglycemia risk. Harmed of the cause on insulin secretion that appears through chronic sulfonylurea organization is due to receptor down-regulation for sulfonylureas on the β-cell surface. This condition vanishes after medication has been stopped for a period of time. In reality, the first administering effect reappears when the administration of these drugs resumes. By a common mechanism, sulfonylureas can induce somatostatin secretion and inhibit glucagon secretion in ÿ and α-cells. Apart from the pancreas β-cells, sulfonylureas also exert their causes on other cells. For example, a rise in insulin receptors occurs on erythrocytes, monocytes, and adipocytes has been seen in patients treated chronically with sulfonylureas [11]. In addition, sulfonylureas also tend to have other causes: they improve the peripheral use of glucose by two mechanisms of action, by raising the sensitivity and number of insulin receptors, and by inducing hepatic gluconeogenesis. However, their important consequence is raised response of β-cells to collectively non-glucose secretagogues and glucose secretagogues of amino acids, which results in more insulin liberated at any concentration of blood glucose. However, this reality should not be underestimated; they may affect the suppression, often the majority of hepatic glucose production in overnight, thus additional reducing the concentration of fasting blood glucose [12].
D. Metformin, Sulfonylureas, Cancer Mortality and Risk
The medicine of choice for the treatment of type 2 diabetes is metformin & sulfonylurea. It is a safe medication, and very low in cost. So, it remains one of the world's most popular prescription drugs. Further, metformin can inhibit the growth of cancer cells in vitro [13]. Hirsch et al. [14] elucidate that sulfonylureas & metformin selectively destroy stem cells of cancer and inhibit the growth of tumors. They also observed metformin's synergistic activity with chemotherapeutic drugs to minimize tumor mass and extend the remission in nude mice. They illustrate that a combined therapy (metformin & sulfonylureas along with doxorubicin) would bring and sustain remission for at least 60 days after removal of the drug. Relapse of the tumor growth was experimental in doxorubicin-treated mice alone after 20 days. Combination therapy, on the other hand, was associated with a sustained recovery, which may indicate a cure. Meta-analysis of metformin and Sulfonylurea and cancer risk in diabetic patients identified a 1/3rd of reduction in metformin users' overall cancer risk and mortality relative to other antidiabetic drugs [15]. In addition, the combined hazard ratios for the occurrence of different cancer sites in metformin users were lower: 0.19 (93 % CI, 0.11–0.41) for hepatocellular cancer, 1.07 (93 % CI, 0.38–1.09) for lung cancer and 1.02 (93 % CI, 0.42 –0.69) for colorectal cancer; [15]. A recent meta-analysis
specifically explains the possible function of metformin and Sulfonylurea in chemoprevention of hepatic cancer. The study suggested that metformin and Sulfonylurea is associated with a 62?crease in the anticipated risk of liver cancer and a 70?crease in the risk of hepatocellular carcinoma (HCC) in patients with type 2 diabetes. The discovery that the hazard, minimize for occurrence of cancer varies for specific sites may be due to the variation in carcinogenesis at various sites. Hyperglycemia and hyperinsulinemia affect the development of the epithelial malignant tumors such as breast, colon, prostate and the pancreas [16]. Such results support metformin's and Sulfonylurea’s anticancer effect, especially since the study was focused on broad population-based data from several nations, including Asians who are typically insulinopenic and lean. While meta-analysis of observational studies indicates a reduction of the risk of cancer in the metformin and Sulfonylurea users by about one third, the findings of a recent systematic review and collaborative metaanalysis of RCT do not bear this definition. [17]. This meta-analysis gathered data from 10 RCTs for cancer incidence and 12 RCTs for all-cause mortality. Relative to comparators, the summary RR for cancer cases in people randomized to metformin was 0.93 (93 % CI: 0.78–1.19, I2= 13 %). Furthermore, the RR description for all-cause mortality in the metformin and Sulfonylurea users was 0.89 relative to comparators (93 % CI: 0.69–1.02). Metformin and Sulfonylurea was not in favor of review of the testing comparing metformin and Sulfonylurea to normal treatment and testing with periods longer than a year. Nevertheless, the confidence spaces were large, and given the comparators, there was a strong clinical heterogeneity between the testing. In fact, insufficient data were available to analyze the individual cancer endpoints. The short follow-up duration of average of the 4.1 years was a further significant constraint. The significant role of metformin and Sulfonylurea in lowering the risk of cancer was evident in most observational studies when the medication was applied for more than 5 years [18]. Since many patients with diabetes require various drugs for optimum metabolic control, the likelihood of drug connections in both clinical testing and observational studies is an important limitation for cancer risk assessment of exact glucose lowering drugs [19, 20].
3. Conclusion
In conclusion, metformin and Sulfonylurea is an effective and safe medication for the low-cost treatment of type 2 diabetes. The current evidence isn't enough to support metformin's and Sulfonylurea’s anticancer effect. Metformin's and Sulfonylurea’s preventive effect can be apparent over a longer period of time or for, for example, insulin patients in other treatment groups. Long-term randomized clinical trial is required to test the hypothesis metformin and Sulfonylurea has an anticancer effect, particularly designed to assess metformin and Sulfonylurea effect on cancer risk.
4. Abbreviation
ABC: ATP-binding cassette
AMP: Adenosine Monophosphate
AMPK: Adenosine Monophosphate Kinase
CREB: Cyclic AMP Response Element Binding
CI: Cancer Incidence
HER2: Human Epidermal Growth Factor Receptor 2
IGF1: Insulin-like Growth Factor 1
LKB1: Liver Kinase B1
mTOR: Mammalian Target of Rapamycin
NBF: Nucleotide-Binding Folds
SUR: Sulfonylureas Receptor
TM: Transmembrane
TMD: Transmembrane Domains
TORC2: Transducer of regulated
CREB activity 2
RR: Risk Ratio
RCT: Randomised Controlled Trial
References
- L. Deng, Z. Gui, L. Zhao, J. Wang, and L. Shen, “Diabetes mellitus and the incidence of colorectal cancer: an updated systematic review and metaanalysis,” Digestive Diseases and Sciences, vol. 57, no. 6, pp. 1576–1585, 2012.
View at Publisher | View at Google Scholar - Kasper JS & Giovannucci E. A meta-analysis of diabetes mellitus and the risk of prostate cancer. Cancer Epidemiology, Biomarkers and Prevention 15 2056–2062, 2006.
View at Publisher | View at Google Scholar - H. Noto, T. Tsujimoto, T. Sasazuki, and M. Noda, “Significantly increased risk of cancer in patients with diabetes mellitus: a systematic review and metaanalysis,” Endocrine Practice, vol. 17, no. 4, pp. 616– 628, 2011.
View at Publisher | View at Google Scholar - Bonovas S, Filioussi K & Tsantes A. Diabetes mellitus and risk of prostate cancer: a meta-analysis. Diabetologia 47 1071–1078, 2004.
View at Publisher | View at Google Scholar - L. M. Berstein, “Modern approach to metabolic rehabilitation of cancer patients: biguanides (phenformin and metformin) and beyond,” Future Oncology, vol. 6, no. 8, pp. 1313–1323, 2010.
View at Publisher | View at Google Scholar - C. J. Bailey and R. C. Turner, “Metformin,” The New England Journal of Medicine, vol. 334, no. 9, pp. 574–579, 1996.
View at Publisher | View at Google Scholar - Derosa G, Tinelli C, Maffioli P. Effects of pioglitazone and rosiglitazone combined with metformin on body weight in people with diabetes. Diabetes Obes Metab.11:1091–9, 2009.
View at Publisher | View at Google Scholar - I.N. Alimova, B. Liu, Z. Fan et al., “Metformin inhibits breast cancer cell growth, colony formation and induces cell cycle arrest in vitro,” Cell Cycle, vol. 8, no. 6, pp. 909–915, 2009.
View at Publisher | View at Google Scholar - A.M. Mulligan, F. P. O’Malley, M. Ennis, I. G. Fantus, and P. J. Goodwin, “Insulin receptor is an independent predictor of a favorable outcome in early stage breast cancer,” Breast Cancer Research and Treatment, vol. 106, no. 1, pp. 39–47, 2007.
View at Publisher | View at Google Scholar - B. Kahn, T. Alquier, D. Carling, and D. G. Hardie, “AMP activated protein kinase: ancient energy gauge provides clues to modern understanding of metabolism,” Cell Metabolism, vol. 1, no. 1, pp. 15–25, 2005.
View at Publisher | View at Google Scholar - Eyileten, C., Kaplon-Cieslicka, A., MirowskaGuzel, D., Malek, L., & Postula, M. Antidiabetic effect of brain-derived neurotrophic factor and its association with inflammation in type 2 diabetes mellitus. Journal of diabetes research, 2017.
View at Publisher | View at Google Scholar - Shorr RI, Ray WA, Daugherty JR, Griffin MR. Individual sulfonylureas and serious hypoglycemia in older people. J Am Geriatr Soc 44: 751-5, 1996.
View at Publisher | View at Google Scholar - S. L. Bowker, S. R. Majumdar, P. Veugelers, and J. A. Johnson, “Increased cancer-related mortality for patients with type 2 diabetes who use sulfonylureas or insulin,” Diabetes Care, vol. 29, no. 2, pp. 254–258, 2006.
View at Publisher | View at Google Scholar - H. A. Hirsch, D. Iliopoulos, P. N. Tsichlis, and K. Struhl, “Metformin selectively targets cancer stem cells, and acts together with chemotherapy to block tumor growth and prolong remission,” Cancer Research, vol. 69, no. 22, article 8832, 2009.
View at Publisher | View at Google Scholar - K. S. Peairs, B. B. Barone, C. F. Snyder et al., “Diabetes mellitus and breast cancer outcomes: a systematic review and meta-analysis,” Journal of Clinical Oncology, vol. 29, no. 1, pp. 40–46, 2011.
View at Publisher | View at Google Scholar - E. Giovannucci, “Metabolic syndrome, hyperinsulinemia, and colon cancer: a review,” The American Journal of Clinical Nutrition, vol. 86, no. 3, pp. 836S–842S, 2007.
View at Publisher | View at Google Scholar - R. J. Stevens, R. Ali, C. R. Bankhead et al., “Cancer outcomes and all-cause mortality in adults allocated to metformin: systematic review and collaborative meta-analysis of randomised clinical testing,” Diabetologia, vol. 55, pp. 2593–2603, 2012.
View at Publisher | View at Google Scholar - DeCensi, M. Puntoni, P. Goodwin et al., “Metformin and cancer risk in diabetic patients: a systematic review and metaanalysis,” Cancer Prevention Research, vol. 3, no. 11, pp. 1451–1461, 2010.
View at Publisher | View at Google Scholar - R. C. Turner, C. A. Cull, V. Frighi, and R. R. Holman, “Glycemic control with diet, sulfonylurea, metformin, or insulin in patients with type 2 diabetes mellitus. Progressive requirement for multiple therapies (UKPDS 49),” Journal of the American Medical Association, vol. 281, no. 21, pp. 2005–2012, 1999.
View at Publisher | View at Google Scholar - R. Ruiter, L. E. Visser, M. P. P. Van Herk-Sukel et al., “Lower risk of cancer in patients on metformin in comparison with those on sulfonylurea derivatives: results from a large population-based follow-up study,” Diabetes Care, vol. 35, no. 1, pp. 119–124, 2012.
View at Publisher | View at Google Scholar - Kasper JS, Giovannucci E. A meta-analysis of diabetes mellitus and the risk of prostate cancer. Cancer Epidemiology and Prevention Biomarkers. 2006 Nov 1;15(11):2056-62.
View at Publisher | View at Google Scholar - Bonovas S, Filioussi K, Tsantes A. Diabetes mellitus and risk of prostate cancer: a meta-analysis.Diabetologia. 2004 Jun 1;47(6):1071-8.
View at Publisher | View at Google Scholar - Groop LC. Sulfonylureas in NIDDM. Diabetes care. 1992 Jun 1;15(6):737-54.
View at Publisher | View at Google Scholar - Nagi DK, Yudkin JS. Effects of metformin on insulin resistance, risk factors for cardiovascular disease, and plasminogen activator inhibitor in NIDDM subjects: a study of two ethnic groups. Diabetes care. 1993 Apr 1;16(4):621-9.
View at Publisher | View at Google Scholar - Alberti KG, Zimmet P, DeFronzo RA. International textbook of diabetes mellitus. J. Wiley; 1997.
View at Publisher | View at Google Scholar - DeFronzo RA. Pathogenesis of type 2 (non-insulin dependent) diabetes mellitus: a balanced overview. Diabetologia. 1992 Apr 1;35(4):389-97.
View at Publisher | View at Google Scholar - Groop L, Widen E, Franssila-Kallunki A, Ekstrand A, Saloranta C, Schalin C, Eriksson J. Different effects of insulin and oral antidiabetic agents on glucose and energy metabolism in type 2 (noninsulin-dependent) diabetes mellitus. Diabetologia. 1989 Aug 1;32(8):599-605.
View at Publisher | View at Google Scholar

 Clinic
Clinic
