Research Article | DOI: https://doi.org/10.31579/2835-835X/126
Saudi Punica granatum L Peel Extract Achievement in Eradicating Zoonotic Pathogens
*Corresponding Author: Sherifa Mostafa M. Sabra S. Const., Prof., Dr. Microbiology.
Citation: Sherifa Mostafa M. Sabra, (2026), Saudi Punica granatum L Peel Extract Achievement in Eradicating Zoonotic Pathogens, Clinical Trials and Case Studies, 5(1); DOI:10.31579/2835-835X/126
Copyright: © 2026, Sherifa Mostafa M. Sabra. This is an open-access artic le distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: 02 February 2026 | Accepted: 11 February 2026 | Published: 18 February 2026
Keywords: punica granatum peel extract; escherichia coli; listeria monocytogenes; candida albicans; staphylococcus aureus; bacillus cereus; clostridium sp; campylobacter sp; shigella sp; brucella sp; salmonella sp
Abstract
Punica granatum peel extract is anti-zoonotic pathogens and energetic role in the zoonotic health field. It contains anti-zoonotic pathogens as phenolic acid, flavonoids, and Tannins. The aim was to conduct a laboratory experiment to test Saudi Punica granatum peel extract against zoonotic pathogens in order to demonstrate the extract's role in zoonotic health. The methodology was for an experiment exposing zoonotic pathogens to Saudi Punica granatum peel extract. The results of group A was included Escherichia coli, Listeria monocytogenes, and Candida albicans, after 12 hours eradications were (72.0%, 72.5% and 70.2%), and after 24 hours were (96.3%, 95.0%, and 95.2%), in range (95%-100%). Group B was included Staphylococcus aureus, and Bacillus cereus, after 12 hours were (60.5%, and 61.2%), and after 24 hours were (90.7%, and 91.8%), in range (90%-95%). Group C was included Clostridium sp., Campylobacter sp., and Shigella sp., after 12 hours were (56.3%, 57.6%, and 57.0%), and after 24 hours were (86.2%, 87.1%, and 86.0%), in range (85%-90%). Group D was included Brucella sp., and. Salmonella sp, after 12 hours were (61.4%, and 58.8%), and after 24 hours were (80.7% and 82.4%), in range (80%-85%). It was concluded that Saudi Punica granatum peel extract was capable to eradicate zoonotic pathogens pose a human's health risk, can be transmitted to humans and cause zoonotic diseases. It also protected animals and animal products, thus possessing cross-health protection properties for both humans and animals. It was recommended that Saudi Punica granatum peel extract using of in appropriate medicinal quantities to maintain zoonotic health.
Introduction
Punica granatum L is under "Subfamily Punicoideae", Punica granatum peel has a good position in the zoonotic health. Punica granatum peel extract is using as anti-zoonotic pathogens and plays an energetic role in the zoonotic health field. Punica granatum peel extract contains anti-zoonotic pathogens as phenolic acid, flavonoids, Tannins etc., that have been found very effective in the zoonotic health field. Due to the presence of anti-zoonotic pathogens action is authoritative for the zoonotic health improvement. The anti-zoonotic function is acting against various zoonotic pathogens [1].
Punica granatum peel extract contains compounds 5-hydroxy-methyl-furfural (36.6%), D-sucrose (23.2%), sorbitol (6.7%), palmitic acid β-mono-glyceride (5.6%), 2-furan-carboxal-dehyde (3.5%) and β-D-gluco-pyranose (3.3%) [2]. Punica granatum peel extract contains substantial quantities of poly-phenols as ellagic tannins, gallic acid, and ellagic acid [3].
Punica granatum peel extract contains active compounds control zoonotic pathogens activities [4]. Punica granatum peel extract contains active mixtures demonstrated as anti-zoonotic pathogens activity [5]. Punica granatum peel extracts due to content of high levels of phenolics and exhibited as antibacterial activity against zoonotic bacteria [6].
Punica granatum peel extract was very effective to control various zoonotic pathogens Staphylococcus sp. [7]. Punica granatum peel extract was effective against zoonotic bacteria Salmonella sp., Escherichia coli, and Staphylococcus aureus, control the production of enterotoxin [8]. Punica granatum peel extract had anti-zoonotic properties against Shigella sp. [7]. Punica granatum peel extract was highly susceptible to zoonotic bacteria Shigella sp. [9]. Punica granatum peel extract had anti-zoonotic pathogens activities Staphylococcus aureus NCTC 6571 [10]. Punica granatum peel extract was preventing zoonotic bacteria Escherichia coli, and Staphylococcus aureus [8]. Punica granatum peel extract had anti-zoonotic bacteria on Escherichia coli, and Listeria monocytogenes [11]. Punica granatum peel extracts had anti-zoonotic activity against Bacillus sp., Staphylococcus aureus, Escherichia coli, and Salmonella sp. Gram-positive bacteria were more sensitive to the extracts than Gram-negative [12]. Punica granatum peel extract used as anti-zoonotic microbial activity against enteropathogenic zoonotic bacteria Escherichia coli, Salmonella sp., and Shigella sp. [13].
Punica granatum peel extract had yeast anti-zoonotic on Candida albicans infection [14]. Punica granatum peel extract had positive anti-zoonotic on Candida albicans from family Saccharomycetaceae [15]. Punica granatum peel extract had preventing various zoonotic pathogens Candida albicans [16].
The importance it was for an aim of the research was to conduct a laboratory experiment to test Saudi Punica granatum peel extract against zoonotic pathogens in order to demonstrate the extract's role in zoonotic health.
Methodology:
Peel Extract:
Saudi Punica granatum sample was bought from the town market, the peel was manually peeled, was washed by distilled water, and 70% ethanol. The Saudi Punica granatum peel was extracted by "Hydro-Distillation" used "Clevenger-Type-Device". The dried peel was crushed by "Sterile-Mixer", was additional 30 g ground powder + 600 mL distilled water in 1000 mL bottle covering 600 mL, the mixture was boiling for 5 hours. Saudi Punica granatum peel extract was available as mL/100 g dry matter, then anhydrous Na2SO4 was dehydrated and dark glass container kept at 4◦C [17].
Zoonotic Pathogens:
The zoonotic pathogens isolates were collected from "Isolated-Lab.", were included Staphylococcus aureus, Clostridium sp., Bacillus cereus, Brucella sp., Salmonella sp., Escherichia coli, Campylobacter sp., Shigella sp., Listeria monocytogenes, and Candida albicans. They were cultured in "Muller-Hinton-Broth", were incubated at 37°C for 18 hours, then were subculture for pure colony on "Muller-Hinton-Agar" at 37°C for 18 hours. Then pure isolates were made in "Muller-Hinton-Broth", (1-2×108 CFU/mL) [18].
Laboratory Test:
An equal quantity of Saudi Punica granatum peel extract and pure isolates suspension were placed in each hole of "Micro-Titration-Plate", and were made three holes for each isolate. The plate was mixed by "Electric-Vibrator." The plate was incubated at (35-37) °C, then was checked at (12 and 24) hours. A mixture loopful was taken from each hole for culture on "Molar-Hinton-Agar" was incubated for 24 hours at (35-37) °C, the colony count was made [11].
Statistical Analyses
The results were made in triple trials, were existed as mean with standard errors. These final results were calculated by "Microsoft Excel Software" [19].
Results and Discussions:
Zoonotic pathogen | Item | Time | Group | ||
Zero hours | 12 hours | 24 hours | |||
Escherichia coli | Colony count | 82±3 | 23±2 | 3±2 | A |
lived percent | 100% | 28.0% | 3.7% | ||
Eradicated percent | 00.0% | 72.0% | 96.3% | ||
Listeria monocytogenes | Colony count | 80±3 | 22±2 | 4±2 | |
lived percent | 100% | 27.5% | 5.0% | ||
Eradicated percent | 00.0% | 72.5% | 95.0% | ||
Candida albicans | Colony count | 84±3 | 25±2 | 4±2 | |
lived percent | 100% | 29.8% | 4.8% | ||
Eradicated percent | 00.0% | 70.2% | 95.2% | ||
Staphylococcus aureus | Colony count | 86±3 | 34±2 | 8±2 | B |
lived percent | 100% | 39.5% | 9.3% | ||
Eradicated percent | 00.0% | 60.5% | 90.7% | ||
Bacillus cereus | Colony count | 85±3 | 33±2 | 7±2 | |
lived percent | 100% | 38.8% | 8.2% | ||
Eradicated percent | 00.0% | 61.2% | 91.8% | ||
Clostridium sp. | Colony count | 87±3 | 38±2 | 12±2 | C |
lived percent | 100% | 43.7% | 13.8% | ||
Eradicated percent | 00.0% | 56.3% | 86.2% | ||
Campylobacter sp. | Colony count | 85±3 | 36±2 | 11±2 | |
lived percent | 100% | 42.4% | 12.9% | ||
Eradicated percent | 00.0% | 57.6% | 87.1% | ||
Shigella sp. | Colony count | 86±3 | 37±2 | 12±2 | |
lived percent | 100% | 43.0% | 14.0% | ||
Eradicated percent | 00.0% | 57.0% | 86.0% | ||
Brucella sp. | Colony count | 83±3 | 32±2 | 16±2 | D |
lived percent | 100% | 38.6% | 19.3% | ||
Eradicated percent | 00.0% | 61.4% | 80.7% | ||
Salmonella sp. | Colony count | 85±3 | 35±2 | 15±2 | |
lived percent | 100% | 41.2% | 17.6% | ||
Eradicated percent | 00.0% | 58.8% | 82.4% | ||
Table 1: An estimated percentage of zoonotic pathogens after contact to Saudi Punica granatum peel extract
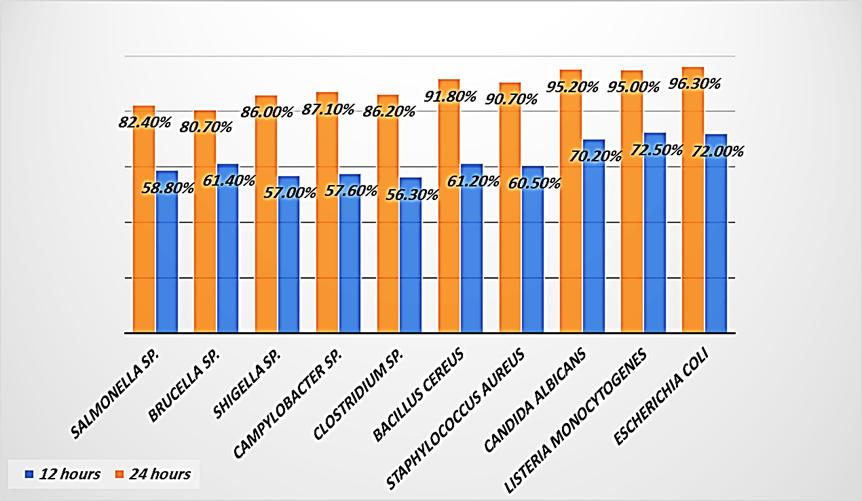
Figure 1: An estimated percentage of zoonotic pathogens after contact to Saudi Punica granatum peel extract
Table (1), and Figure (1) were showed an estimated percentage of zoonotic pathogens after contact to Saudi Punica granatum peel extract. Because Saudi Punica granatum peel extract contains active compounds that eradicated zoonotic pathogens [1-6].
Group A were contained zoonotic pathogens included Escherichia coli, Listeria monocytogenes, and Candida albicans. The results after twelve hours were showed the eradicated percent were (72.0%, 72.5% and 70.2%), and the results after twenty-four hours were showed the eradicated percent were (96.3%, 95.0%, and 95.2%). The results indicated that Saudi Punica granatum peel extract was eradicated group A in a range of (95%-100%) for zoonotic pathogens [8, 11-16].
Table (1), and Figure (1) were showed group B were contained zoonotic pathogens included Staphylococcus aureus, and Bacillus cereus. The results after twelve hours were showed the eradicated percent were (60.5%, and 61.2%), and the results after twenty-four hours were showed the eradicated percent were (90.7%, and 91.8%). The results indicated that Saudi Punica granatum peel extract was eradicated group B in a range of (90%-95%) for zoonotic pathogens [7-8, 10-12].
Table (1), and Figure (1) were showed group C were contained zoonotic pathogens included Clostridium sp., Campylobacter sp., and Shigella sp. The results after twelve hours were showed the eradicated percent were (56.3%, 57.6%, and 57.0%), and the results after twenty-four hours were showed the eradicated percent were (86.2%, 87.1%, and 86.0%). The results indicated that Saudi Punica granatum peel extract was eradicated group C in a range of (85%-90%) for tested zoonotic pathogens [9, 11, 13].
Table (1), and Figure (1) were showed group D were contained zoonotic pathogens included Brucella sp., and. Salmonella sp. The results after twelve hours were showed the eradicated percent were (61.4%, and 58.8%), and the results after twenty-four hours were showed the eradicated percent were (80.7% and 82.4%). The results indicated that Saudi Punica granatum peel extract was eradicated group D in a range of (80%-85%) for zoonotic pathogens [11-13].
The results were showed the main components effects in Saudi Punica granatum peel extract had the property of zoonotic pathogens eradication that are present in the animal environment and can be transmit to humans. These affecting the shared environment and causing zoonotic humans' diseases, especially farmers who are in close proximity to animals or workers handle animal products [1-6].
Conclusions:
It was concluded that, the research had shown that Saudi Punica granatum peel extract was capable of eradicate zoonotic pathogens pose a human's health risk, as these zoonotic pathogens can be transmitted to humans and cause zoonotic diseases. It also protected animals and animal products, thus possessing cross-health protection properties for both humans and animals.
Recommendations:
It was recommended that the using of Saudi Punica granatum peel extract in appropriate medicinal quantities to maintain zoonotic health.
Acknowledgments:
Thanks, were sent to all researchers who contributed to this research.
References
- Saifulazmi, F., Rohani, R., Harun, S., Bunawan, H., Hamezah, S., et al. (2022). A review with updated perspectives on the antiviral potentials of traditional medicinal plants and their prospects in antiviral therapy. Life, 12(8): 1287.
View at Publisher | View at Google Scholar - Abdel-Aty, S., Ahmed, M., Desheesh, A., & El-Kashef, M. et al. (2025). Punica granatum Peel as Antifungal and Antibacterial Source. Egyptian Journal of Chemistry, 68(6): 15-25.
View at Publisher | View at Google Scholar - Caminiti, R., Mazza, V., Maiuolo, J., Liuzzi, F., Oppedisano, F., et al. (2026). The Significant Antioxidant Effect Exerted by Pomegranate (Punica granatum): The Hidden Polyphenols. Antioxidants, 15(3): 276.
View at Publisher | View at Google Scholar - Gosset-Erard, C., Zhao, M., Lordel-Madeleine, S., & Ennahar, S. et al. (2021). Identification of punicalagin as the bioactive compound behind the antimicrobial activity of pomegranate (Punica granatum L.) peels. Food chemistry, 352, 129396.
View at Publisher | View at Google Scholar - Silva, A., Vidal, S., Filho, A., Agatão, M., Berbert, C., et al. (2025). Rapid bactericidal activity of Punica granatum L. peel extract: a natural alternative for mastitis prevention in dairy cattle. Molecules, 30(11): 2387.
View at Publisher | View at Google Scholar - Kiran, S., Tariq, A., Iqbal, S., Naseem, Z., Siddique, W., et al. (2024). Punicalagin, a pomegranate polyphenol sensitizes the activity of antibiotics against three MDR pathogens of the Enterobacteriaceae. BMC complementary medicine and therapies, 24(1): 93.
View at Publisher | View at Google Scholar - Rasheed, M., Riaz, A., Asad, M., Mumtaz, R., Ehsan, S., et al. (2023). And Shabeer I, 2023. Efficacy of natural products against zoonotic disorders. Zoonosis, Unique Scientific Publishers, Faisalabad, Pakistan, 1, 541-564.
View at Publisher | View at Google Scholar - Saleh, R. O., Najim, A., Mukhlif, Y., & Jasim, A. et al. (2025). Assessing the Effect of Pomegranate Peel Extract against Escherichia coli and Staphylococcus aureus Isolated from Pregnant Women Suffering from Urinary Tract Infections in Al-Ramadi. Medical Journal of Babylon, 22(4): 1501-1508.
View at Publisher | View at Google Scholar - Ahmed, F., Hussien, Q., & Hassan, I. et al. (2025). Punica Granatum Bioactive Activity Against Gram Negative Isolates from Post-Surgical Infections in Erbil Province. The Review of Diabetic Studies, 36-49.
View at Publisher | View at Google Scholar - Farhat, G., Cheng, L., Al-Dujaili, A., & Zubko, M. et al. (2024). Antimicrobial potential of pomegranate and lemon extracts alone or in combination with antibiotics against pathogens. International Journal of Molecular Sciences, 25(13): 6943.
View at Publisher | View at Google Scholar - Shleghm, R., Mansoor, A., & Naeemah, C. et al. (2024). Evaluation of the antibacterial effects of the Punica granatum peels extracts against some pathogenic bacteria: An in vivo and in vitro study. Biomedical and Pharmacology Journal, 17(3): 2065-2070.
View at Publisher | View at Google Scholar - Mendes, M., Gomes Fontoura, M., Rodrigues, S., Souza, S., et al. (2023). Therapeutic potential of Punica granatum and isolated compounds: Evidence‐based advances to treat bacterial infections. International Journal of Microbiology, 2023(1): 4026440.
View at Publisher | View at Google Scholar - Wu, W., Mis Solval, K., & Chen, J. et al. (2025). Inhibition of Salmonella enterica and enterohemorrhagic Escherichia coli by ethanolic extracts of pomegranate peels. Microbiology Research, 16(1): 13.
View at Publisher | View at Google Scholar - Ferreira, S., Moreno, C., de Souza Duarte, E., Moreira, G., Ucella-Filho, et al. (2025). Exploring the antifungal potential and action mechanism of pomegranate peel extract against Candida species in planktonic and biofilm conditions. Microbial Pathogenesis, 204, 107596.
View at Publisher | View at Google Scholar - Mandal, K., Katuwal, S., Tettey, F., Gupta, A., Bhattarai, S., et al. (2022). Current research on zinc oxide nanoparticles: synthesis, characterization, and biomedical applications. Nanomaterials, 12(17): 3066.
View at Publisher | View at Google Scholar - El-Hamid, A., Ibrahim, D., Abdelfattah-Hassan, A., Mohammed, B., Pet, I., et al. (2024). Silver nanoparticles loaded with pomegranate peel extract and hyaluronic acid mediate recovery of cutaneous wounds infected with Candida albicans. Frontiers in Cellular and Infection Microbiology, 14, 1469493.
View at Publisher | View at Google Scholar - Elkiran, O., & Telhuner, O. (2025). Chemical Profiles and Antimicrobial Activities of Essential Oil from Different Plant Parts of Fennel (Foeniculum vulgare Mill.). Food Science & Nutrition, 13(5): e70307.
View at Publisher | View at Google Scholar - Kovařovic, V., Sedláček, I., Petráš, P., Králová, S., Mašlaňová, I., et al(2022). Staphylococcus ratti sp. nov. isolated from a lab rat. Pathogens, 11(1): 51.
View at Publisher | View at Google Scholar - Kiviaho, K., Anttonen, M., Alfthan, H., & Itkonen, O. (2025). Reliable quantification of fecal elastase-1: A study on sample stability, IDK ELISA and IDK Extract® device. Clinica Chimica Acta, 120541.
View at Publisher | View at Google Scholar

 Clinic
Clinic
