Case report | DOI: https://doi.org/10.31579/2834-796X/055
Neonatal Ebsteins Anomaly presenting with unique features: Rare case report
Professor and Head of the department of Cardiology, JLN Medical College, Ajmer, Rajasthan (India) Govind Mittal, MSc (Economics)
*Corresponding Author: Akhil Mehrotra, Chief, Pediatric and Adult Cardiology, Prakash Heart Station, Nirala Nagar, Lucknow, UP, India.
Citation: Akhil Mehrotra, Mohammad Shaban, Faiz Illahi Siddiqui, (2024), Neonatal Ebsteins Anomaly presenting with unique features: Rare case report, International Journal of Cardiovascular Medicine, 3(1); DOI:10.31579/2834-796X/055
Copyright: © 2024, Akhil Mehrotra. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 25 January 2024 | Accepted: 13 February 2024 | Published: 29 February 2024
Keywords: ebsteins anomaly; double-outlet left ventricle; congenital heart disease; neonatal ebsteins anomaly; tricuspid regurgitation
Abstract
Ebstein’s anomaly (EA) of the tricuspid valve is an uncommon malformation, representing an estimate 0.3% to 0.6% of all cases of congenital heart disease. The basic defect involves a deformed and displaced tricuspid valve, resulting in symptoms that vary with age of the patient and severity of the defect. Symptoms are referable to progressive heart failure and cyanosis resulting from severe tricuspid insufficiency. Once symptoms appear the only treatment for Ebstein’s anomaly is surgical intervention.
Infants with symptomatic Ebstein’s anomaly have a poor prognosis, with an expected mortality rate of 50% to 75%. Previous attempts at palliative surgical treatment in this neonatal group have produced equally dismal results.
Herein, we are reporting a very sick 3 day old neonate afflicted with Ebsteins anomaly with double-outlet left ventricle (DOLV) associated with myriads of other significant congenital heart defects.
Introduction
Ebstein’s anomaly is a rare congenital heart disorder occurring in ≈1 per 200,000 live births and accounting for <1>
Ebstein’s anomaly is characterized by various degrees of inferior displacement of the proximal attachments of the tricuspid valve (TV) ring, TV dysplasia, right ventricular dysplasia, and abnormalities of the distal attachments of the TV (Figures 1-4).

Figure 1: Diagrammatic illustration of a heart with Ebstein's anomaly highlighting the anatomical abnormalities. The right atrium (RA) and tricuspid annulus is dilated. The septal tricuspid leaflet is apically displaced (arrow). The coronary sinus (CS) ostium is dilated, AV node (AVN) is irregular. ASD, atrial septal defect; aRV, atrialised RV; LA, left atrium; LV, left ventricle; RV, right ventricle.

Figure 2: Diagram of the "displacement index." The distance from the hinge point of the anterior mitral leaflet is measured to the hinge point of the delaminated septal tricuspid leaflet. This measurement divided by the body surface area equals the displacement index. A displacement index >8 mm/m2 is diagnostic of Ebstein anomaly. A small functional right ventricle (RV) is present inferior to the coaptation point of the tricuspid valve. RA, right atrium; TVA, tricuspid valve annulus.

Figure 3: Severe Ebstein anomaly: pathology specimen aRV, atrialized right ventricle
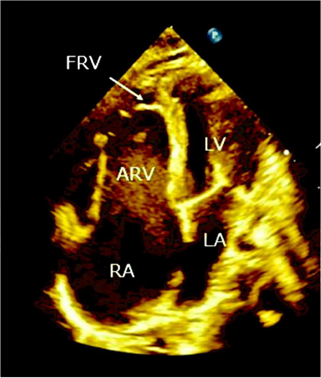
Figure 4: Transthoracic echocardiography apical four chamber view. RA, right atrium; LA, left atrium; ARV, atrialized right ventricle; FRV, functional right ventricle.
These morphologic deformities, in widely different degrees of severity, are associated with a variety of hemodynamic alterations leading to cyanosis, congestive heart failure, and arrhythmias. Refinement of tricuspid valvuloplasty and plication techniques [3-4] has opened the way to a satisfactory long-term outlook for the majority of older children and adults, who generally are only mildly or moderately symptomatic. Ebstein’s anomaly presenting in neonates and young infants, however, has considerably less favorable natural history with a reported mortality rate of as high as 75% [5-6].
Double-outlet ventricles with concordant AV connection account for 1% of all cases of congenital heart disease, and DOLV accounts for <5>
The diagnosis can be made in uterus by echocardiography and if the gestational age is <24>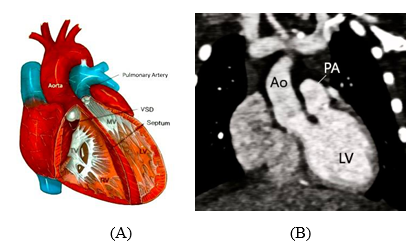
Figure 5: (A) Diagram of the most common form of DOLV showing situs solitus and AV concordance, rightward/anterior or right/lateral Ao with subvalvar or valvar pulmonary stenosis. MV, Mitral valve; TV, tricuspid valve. (B) CT angiography. An oblique coronal multiplanar reformatted image shows the Ao and PA in side-by-side orientation, originating from the LV. More than 50% of the Ao is seen arising from the LV.
It is apparent that isolated neonatal Ebsteins anomaly or DOLV is a dreaded presentation and coexistence of EA alongwith DOLV in a newborn could be furthermore disastrous and lethal. To our knowledge only scarce case reports of EA in association with DOLV have been published earlier [11-13], and all of these patients had a highly malignant course of illness with early mortality.
Case Report
A 3 day old female neonate was referred to us for a comprehensive cardiovascular evaluation.
She was a full term delivery by Caesarean section and was delivered at a private hospital, from a primipara woman of 21 years of age. There was no history of maternal risk factors of CHD (obesity, diabetes, febrile illness, smoking, alcohol intake, teratogenic drug use, or radiation exposure). On clinical examination, the child was very “sick-looking” and was in congestive heart failure as evidenced by the presence of respiratory distress, intercostal retractions, tachypnea, facial edema and swelling in all the four extremities (Figure 6 A-D).

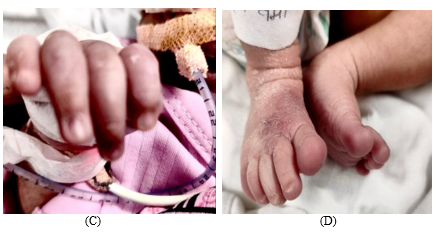
Figure 6: (A) Facial edema, (B) Pectus excavatum, intercostal retractions, (C) Cyanosis of fingers, (D) Cyanosis of toes.
She was of average built, highly irritable and persistently crying. Her weight was 3.7 kg, respiratory rate was 38/min, pulse rate was 98/mm, blood pressure was 80/60 mmHg, and SPO2 was 65% at room air. The child was cyanosed with bluish coloration of tongue, lips, all the fingers, and toes. There was a typical pectus excavatum deformity of the chest without any other musculoskeletal anomalies. All the peripheral pulses were normally palpable without any radio femoral delay. Rest of the systemic examination was unremarkable.
On cardiovascular examination there was presence of Grade 2/6 pansystolic murmur over precordium, best head over lower left sternal border.
Xray chest (AP view) (Figure 7) revealed massive cardiomegaly with a cardiothoracic ratio of 0.78. Moreover, there was severely diminished pulmonary blood flow.
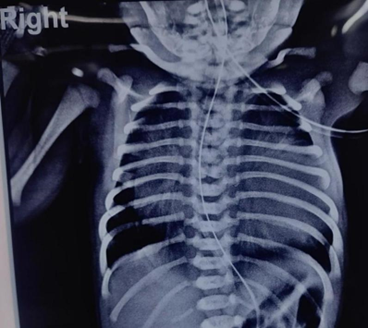
Figure 7: Xray chest A-P view: massive cardiomegaly with cardiothoracic ratio of 0.78 and pulmonary oligemia.
ECG exhibited (Figure 8) sinus tachycardia with a ventricular rate of 98/mm, partial RBBB with a right axis deviation, and ”Himalayan” P waves.
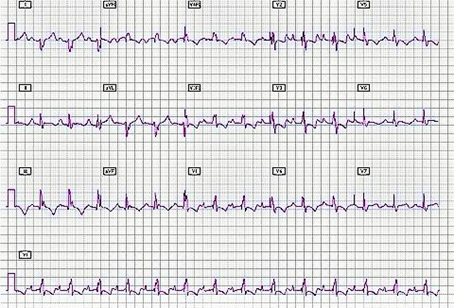
Figure 8: Resting ECG shows sinus tachycardia, partial RBBB, right axis deviation and “Himalayan” P waves.
Transthoracic color doppler echocardiography (Figures 9-14)
Standard transthoracic color doppler echocardiography was performed by the author in the classical subcostal, parasternal long axis (LX), parasternal short axis (SX), 4-Chamber (4CH), 5-Chamber (5CH) and suprasternal views. The echocardiographic characteristics of the neonate are outlined:
(I) Levocardia
Situs Solitus
Concordant D-Bulboventricular Loop
AV Concordance
DOLV – Both great arteries are arising from LV with D-malposition of great arteries.
Left Aortic Arch
Confluent Pulmonary Arteries.
Normal pulmonary venous drainage.
Normal systemic venous drainage.
(II) EBSTEIN’S ANOMALY - (Carpentier Type D)
- TRICUSPID REGURGITATION (Severe)
- Septal leaflet of TV is apically displaced.
- Tricuspid septal leaflet displacement of 13 mm from the mitral insertion.
- The anterior leaflet is large and immobile with the tips adhered to the lateral wall of RV.
- TV orifice is displaced downwards into the RV cavity.
- Low velocity TR jet present (TR velocity 1.69 m/sec).
- On color flow mapping TR jet area 4.15 sq.cm; occupying 45 % of RA area, central jet .
(III) ATRIAL SEPTAL DEFECT (Large)
Ostium secundum type
Size 6.3 mm
Rt. to Lt. Shunt.
Superior and inferior rims of ASD are flail and hyper mobile.
(IV) DOUBLE OUTLET LEFT VENTRICLE
- Both great arteries are arising from left ventricle.
(V) D-MALPOSITION OF GREAT ARTERIES.
- Aorta is anterior and to the right of pulmonary artery
- Pulmonary artery is posterior and to the left of Aorta
(VI) ATRESIA OF THE PULMONARY VALVE
- Severe hypoplasia of branch pulmonary arteries. MPA(D) 3.0 mm
MPA(D) 3.0 mm
LPA(D) 2.2 mm
RPA(D) 2.3 mm
- A thin solitary aorta-pulmonary collateral was visualized connecting descending aorta to the left pulmonary artery with left to Rt shunt.
(VII) Huge RA (Atrialized RV)
Dilated RV
Normal biventricular systolic function.
Normal LVEF = 50 %
No regional wall motion abnormality present.
(VIII)No evidence of ventricular septal defect, coarctation of aorta or bicuspid aortic valve.
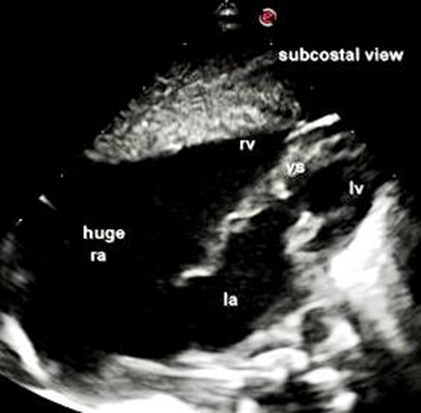
Figure 9: Subcostal View shows dilated RV and huge RA.

Figure 10: LX View exhibits dilated RV and small LV.
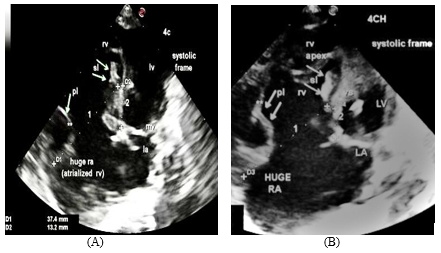
Figure 11: (A) 4CH View(systolic frame) displays normal sized LV, LA, dilated RV, Huge RA( Atrialized RA), and solitary oblique arrow denotes large anterior leaflet adhered to the lateral right ventricular free wall , dilated TV annulus is measuring 37.4 mm ( designated as 1 ), sl and two oblique arrows indicate rudimentary septal leaflet of TV ,tricuspid septal leaflet displacement from mitral insertion is 13.2 mm( designated as 2), which is consistent with severe grade of Ebsteins anomaly- ( Carpentier Type D). (B) 4CH View (systolic frame)- this image represents an enlarged view of figure 11 (A).
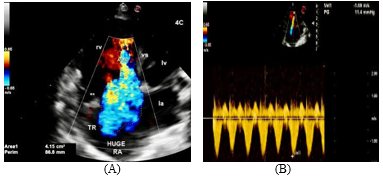
Figure 12: (A) 4CH view. On color flow mapping a severe tricuspid regurgitation (TR) jet is visualised. (B) On continuous wave Doppler analysis across TV, a low velocity signal of TR jet is delineated ( TR velocity 1.69 m/sec ).
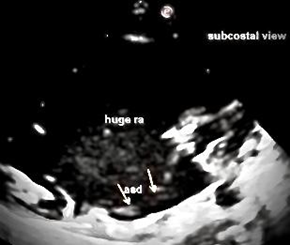
Figure 13: Subcostal View exhibits a large ostium secundum ASD with huge RA.
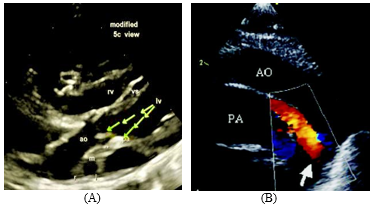
Figure 14: (A) Modified 5CH View clearly delineating Double Outlet Left Ventricle with D- malposition of great arteries- (aorta is anterior and to the right of pulmonary artery) with atresia of the pulmonary valve (designated as pa and **) with hypoplasia of main, left and right pulmonary arteries, m, main pulmonary artery, l, left pulmonary artery, r, right pulmonary artery. (B) Suprasternal View. A solitary aorto-pulmonary collateral is delineated by thick arrow.
The detailed transthoracic echocardiography illustrates an extremely rare and lethal combination of neonatal Ebsteins Anomaly, DOLV, large ASD with right to left shunt, atresia of the pulmonary valve, hypoplatic branch pulmonary arteries, severe TR with advanced heart failure and hypoxia. Hence, the patient’s parents were advised for admission and management of the child at a tertiary care pediatric cardiovascular institution.
Discussion
After deep search of the literature, only a handful of cases of EA accompanying with DOLV could be encountered [11-13]. Chang et al [11] reported a case of Ebsteins anomaly with DOLV, subaortic VSD and pulmonary stenosis in association with esophageal atresia with tracheo-esophageal fistula (so-called VACTERL association) in a neonate. The patient underwent Blalock-Taussig shunt. Bharati et al [12] mentioned in their series of DOLV that there was only one autopsied case of associated Ebsteins anomaly. In that remarkable case the aorta emerged completely from the left ventricle over a well developed conus. The pulmonary trunk overrode the septum over a posteriorly placed VSD, but emerged mostly from the left ventricle. The aorta was anterior and to the right of the pulmonary trunk and the latter was related to the mitral valve. The VSD was related to the tricuspid but not the mitral valve. This case was associated with fetal coarctation and PDA. Van Praagh et al [13] narrated in their series of 109 cases of DOLV; only one autopsy case, presented with DOLV, sub aortic, VSD, pulmonary stenosis and Ebsteins anomaly.
It is noteworthy that all of these patients of DOLV coexisting with EA had a malignant course of illness, leading to very early mortality.
Ebsteins Anomaly- classification
There are numerous classifications of EA put forward by multiple authors. However, the Carpentier [3], Celermajer [14] classification and GOSE score [15] are currently the most illuminating models for classifying the grades of severity and assessment of mortality risk.
Carpentier Classification
According to the classification of Carpentier [3], EA was divided into four types (Figure 15).
- Type A: Mild apical displacement of the tricuspid valve leaflets with the adequate functional right ventricle.
- Type B: Moderate apical displacement of the tricuspid leaflets with a moderate reduced size but adequate functional right ventricular volume with freely mobile anterior leaflet.
- Type C: Severe apical displacement of the tricuspid valve leaflets with a small functional right ventricle. Anterior leaflet movement is restricted due to abnormal chordal attachments that cause right ventricular outflow tract obstruction.
- Type D: Complete non-delamination of the tricuspid valve leaflets with almost complete atrialization of the right ventricle, only infundibular portion of the right ventricle remaining: "Tricuspid sac".

Figure 15: Carpentier Classification
Celermajer Classification and GOSE score
The Celermajer classification of EA [14] was according to echocardiographic measurements calculating the ratio of the combined area of the right atrium and aRV to that of the fRV and the left heart in a four-chamber view at the end diastole (GOSE = RA + aRV/fRV + LV + LA). This is an echocardiographic grading score for neonates with Ebstein’s
anomaly, The Great Ormond Street Echocardiography (GOSE) score, with grades 1 to 4 [15] (Figure 16). Increasing severity, that is, a higher grade, was associated with a high mortality rate. This classification is particularly helpful with neonatal Ebstein’s anomaly. GOSE score and the corresponding mortality rate are demonstrated in the Table 1.
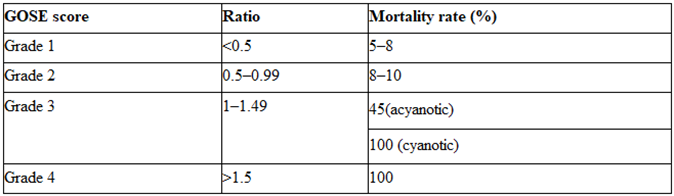
Table 1: GOSE score and mortality rate

Figure 16: GOSE score. RA, right atrium; aRV, atrialized right ventricle; LA, left atrium; LV, left ventricle.
In accordance with Carpentier [3], Celermajer [14] classification and GOSE score [15], our patient is in:
- Carpentier Type D, because of the presence of anterior tricuspid leaflet adhered to the lateral wall of RV alongwith severe tricuspid regurgitation.
- Celemajer classification and GOSE score being-
GOSE score 4
Ratio >1.5
Mortality rate 100 %
We have summarized the characteristic features of 3 case reports of EA coexisting with DOLV encountered in the literature (Table 2).
| Author | Year | Autopsy/Anatomical/Echo diagnosis | Outcome |
| Bharati et al [12] | 1978 | EA, DOLV, D-malposition, VSD related to TV, COA, PDA | Early mortality |
| Van Praagh et al [13] | 1989 | EA, DOLV, sub aortic VSD, PS | Early mortality |
| Chang et al [11] | 2011 | EA, DOLV, subaortic VSD, PS, esophageal atresia with tracheo-esophageal fistula (VACTERL association) | B-T shunt |
B-T; Blalock Taussing, VSD; ventricular septal defect, PS; pulmonary stenosis, COA; coarctation of aorta, PDA; patent ductus arteriosus.
Table 2: Summary of case reports of Ebsteins anomaly coexisting with Double-outlet left ventricle
In contrast to the other 3 previously described case reports, in our patient VSD was absent. Moreover the neonate had atresia of the pulmonary valve with solitary aorto-pulmonary collateral feeding the hypoplastic pulmonary arteries. We later came to know about the unfortunate early demise of the newborn at the age of 10 days, because of numerous complications of the complex disease process.
Celemajer et al [15] reviewed the presentation and outcome of 50 patients with neonatal Ebstein's anomaly seen from 1961 to 1990. The majority (88%) presented in the 1st 3 days of life; cyanosis (80%) was the most common presenting feature. Associated defects, present in 27 infants (54%), included pulmonary stenosis in 11 and atresia in 7. Nine patients (18%) died in the neonatal period; there were 15 late deaths (due to hemodynamic deterioration in 9, sudden death in 5 and a noncardiac cause in 1) at a mean age of 4.5 years (range 4 months to 19 years). Actuarial survival at 10 years was 61%.
According to the GOSE criteria, cardiac death occurred in 0 of 4 infants with grade 1, 1 (10%) of 16 with grade 2, 4 (44%) of 9 with grade 3 and 5 (100%) of 5 with grade 4. In a multivariate analysis of clinical and investigational features at presentation, echocardiographic grade of severity was the best independent predictor of death [15]. Augmented cardiothoracic ratio, presence of associated defects and increasing severity of GOSE score was significantly associated with death in a univariate analysis model [15].
Our case was afflicted with severe grade of Ebsteins anomaly- Carpertier Type D with a GOSE score of expired at the age of 10 days, as predicted by the GOSE criteria of Celermajer et al [15].
Conclusion
The natural history of neonatal Ebsteins Anomaly is extremely variable, although symptomatic presentation in the neonatal period has been associated with a high mortality rate of ≈ 50% [16]. Prior studies suggest that outcome in neonates may be related to the degree of tricuspid valve displacement, severity of tricuspid regurgitation, cardiothoracic ratio, associated cardiac defects and oxygen saturation. Management of neonates with Ebstein's anomaly may therefore be based on the knowledge of echocardiographic grade and the presence or absence of associated defects. Most neonates with grade 1 or 2 disease and no associated defects will survive the neonatal period with supportive treatment only and have a good prognosis; those with associated defects may require surgery for these and may also expect a good outcome. Neonates with grade 3 or 4 disease have a much worse outlook; many die in early life, and tricuspid valve surgery may not alter the poor prognosis. Those who do survive the 1st month of life must have careful clinical follow-up, with particular attention to left ventricular function and cardiac rhythm.
References
- Frescura C, Angelini A, Daliento L, Thiene G. (2000). Morphological aspects of Ebstein’s anomaly in adults. Thorac Cardiovasc Surg. 48: 203–208.
View at Publisher | View at Google Scholar - Edwards WD. (1993). Embryology and pathologic features of Ebstein’s anomaly. Prog Pediatr Cardiol. 2: 5–15.
View at Publisher | View at Google Scholar - Carpentier A, Chauvaud S, Macé L. Relland J. Mihaileanu S, Marino JP, Abry B, Guibourt P. A new reconstructive operation for Ebstein's anomaly of the tricuspid valve. J Thorac Cardio- vasc Surg 1988:96:92-101.
View at Publisher | View at Google Scholar - Quaegebeur JM, Sreeram N, Fraser AG, Bogers AJJC, Stümper OFW, Hess J, Bos E, Sutherland GR. (1991). Surgery for Ebstein's anomaly: The clinical and echocardiographic evaluation of a new technique. J Am Coll Cardiol 17:722-8.
View at Publisher | View at Google Scholar - Radford DJ, Graff RF, Neilson GH. (1985). Diagnosis and natural history of Ebstein's anomaly. Br Heart J 54:517-22.
View at Publisher | View at Google Scholar - Roberson DA, Silverman NH. (1989). Ebstein's anomaly: Echocardio- graphic and clinical features in the fetus and neonate. J Am Coll Cardiol 14:1300-7.
View at Publisher | View at Google Scholar - Menon S,Hagler D.J. (2008). Double-outlet left ventricle: diagnosis and management. Curr Treat Options Cardiovasc Med. 10: 448-452.
View at Publisher | View at Google Scholar - Tchervenkov C.I., Walters III H.L., Chu V.F. (2000). Congenital heart surgery nomenclature and database project: double outlet left ventricle. Ann Thorac Surg. 69: 264-269.
View at Publisher | View at Google Scholar - Allan LD, Crawford DC, Anderson RH, Tynan M. (1985). Spectrum of congenital heart disease detected echocardiographically in prenatal life. Br Heart J. 54:523-6.
View at Publisher | View at Google Scholar - Sunder A, Mohanty B, Sahoo MK. (2013). Double-outlet left ventricle: A rare case. J Family Med Prim Care. 8:1769-71.
View at Publisher | View at Google Scholar - Chang HK, Wang JN, Hung WP, Yen WL, Wu JM. (2011). Double-Outlet Left Ventricle with Ebstein Anomaly in a Neonate with VACTERL Association. Acta Cardiologica Sinica. 27:65-67.
View at Publisher | View at Google Scholar - Bharati S, LEV M, Stewart R, Mcallister Jr HA, Kirklin JW. (1978). The Morphologic Spectrum of Double Outlet Left Ventricle and Its Surgical Significance. Circulation. 58:558-565.
View at Publisher | View at Google Scholar - Van Praagh R, Weinberg PM, Srebro JP. (1989). Double-outlet left ventricle. In: Adams FH, Emmanouilides GC, Riemenschneider JA, eds. Moss and Adams’ Heart Disease in Infants, Children and Adolescents. 4th ed. Baltimore: Williams & Wilkins, 461-85.
View at Publisher | View at Google Scholar - Celermajer DS, Bull C, Till JA, Cullen S, Vassillikos VP, Sullivan ID, Allan L, Nihoyannopoulos P, Somerville J, Deanfield JE. (1994). Ebstein’s anomaly: Presentation and outcome from fetus to adult. Journal of the American College of Cardiology. 23:170-176.
View at Publisher | View at Google Scholar - Celermajer DS, Cullen S, Sullivan ID, Spiegelhalter DJ, Wyse RK, Deanfield JE. (1992). Outcome in neonate with Ebstein’s anomaly. J Am Coll Cardiol. 19:1041-1046.
View at Publisher | View at Google Scholar - Lee LA, Kulik TJ, Sandhu SK, Goldberg CS, Mosca RS, Bove EL, and Charpie JR. (1999). Prognostic Stratification of Neonatal Ebstein’s Anomaly. Pediatric Research. 45:26.
View at Publisher | View at Google Scholar

 Clinic
Clinic
