Research Article | DOI: https://doi.org/10.31579/2835-8295/156
Leio Cmyosarcoma of Urinary Bladder: an Update
*Corresponding Author: Anthony Kodzo-Grey Venyo, Department of Urology, North Manchester General Hospital, United Kingdom.
Citation: Suresh. K, (2026), Leio Cmyosarcoma of Urinary Bladder: an Update, International Journal of Clinical Reports and Studies, 5(2); DOI:10.31579/2835-8295/156
Copyright: © 2026, Anthony Kodzo-Grey Venyo. This is an open-access artic le distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: 27 January 2026 | Accepted: 23 March 2026 | Published: 03 April 2026
Keywords: primary leiomyosarcoma; urinary bladder; rare; biopsy; histopathology; immunohistochemistry; muscle-specific-action; cystectomy; early detection
Abstract
Leiomyosarcomas of the urinary bladder are rare smooth muscle tumours which constitute one per cent (1%) of all urinary bladder malignancies. Even though uncommon, leiomyosarcoma is a clinically significant disease, which usually manifests as a high-grade advanced malignancy with associated substantial morbidity and mortality, if not treated early. The diagnosis of primary leiomyosarcoma of the urinary bladder could be delayed because of the asymptomatic manifestation until the tumour reaches an advanced stage. Pathological examination of specimens of primary leiomyosarcoma of the urinary bladder is highly cellular with infiltrative, interlacing fascicles of spindle cells and associated mitosis, cellular atypia and necrosis. Immunohistochemistry studies of specimens of primary leiomyosarcoma of the urinary bladder does demonstrate tumour cells that exhibit positive staining of muscle-specific-actin, desmin, and caldesmon; with negative staining for ALK-1, EMA, Cytokeratins and GATA3. Other spindle cell lesions including, leiomyoma, inflammatory myofibroblastic tumor and sarcomatoid carcinoma need to be excluded based upon pathology examination of specimens of leiomyosarcoma based upon their immunohistochemistry staining features that are different from the staining features of leiomyosarcoma of the urinary bladder because they represent the differential diagnoses, that need to be excluded by their morphology and immunohistochemistry examination features. All clinicians need to be aware of the occurrence of leiomyosarcomas, which could, on rare occasions, afflict the urinary bladder. Even though rare, primary leiomyosarcomas of the urinary bladder generally portend a poor outcome. The differential diagnosis of spindle cell lesions is extensive, including close benign and malignant mimics, the treatment of all being very different. Immediate radical cystectomy has been associated with longer survival rates for leiomyosarcoma. Leiomyosarcoma, being aggressive, with an associated high recurrence rate and metastatic potential, an early correct diagnosis would aid in the institution of proper management.
Introduction
Non-epithelial tumours of the urinary bladder account for less than five per cent (5%) of overall malignancies of the urinary bladder, with leiomyosarcoma constituting 0.1% of urinary bladder cancers. By 2013, there were more than (>)100 cases reported in the medical literature as leiomyosarcoma, and a total of 192 cases considering all cases of urinary bladder sarcoma. [1] There has been no consensus opinion regarding the standard treatment, and because of the very low incidence, and little is known about the natural history and prognosis of the tumour. Leiomyosarcomas are iterated to portend a poor prognosis, especially if they are of high-grade and stage tumours. [2] In view of the rarity of leiomyosarcoma of the urinary bladder, it would be envisaged that the majority of clinicians all over the world would not have encountered any case of primary leiomyosarcoma of the urinary bladder before, and they might not be familiar with the manifestations and diagnostic features of primary leiomyosarcoma of the urinary bladder. The following article is divided into two parts: (A) Overview of leiomyosarcoma and (B) miscellaneous narrations and discussions from some case reports, case series, and studies related to leiomyosarcoma of the urinary bladder.
Aim
To provide an update on leiomyosarcoma of the urinary bladder.
Method
Internet databases were searched, including: Goggle; google scholar; yahoo; and PUBMED. The search words that were used included: Leiomyosarcoma of urinary bladder; vesical leiomyosarcoma. Fifty-three (53) references were identified, which were used to write the article, which has been divided into two parts: (A) Overview of leiomyosarcoma and (B) miscellaneous narrations and discussions from some case reports, case series, and studies related to leiomyosarcoma of the urinary bladder.
[A] overview
Results
[a] overview
Definition / general iteration
- Leiomyosarcoma is reported to be a malignant mesenchymal tumour with smooth muscle differentiation [3]
Epidemiology
The epidemiology of leiomyosarcoma of the testis and para-testis has been summarised as follows: [3]
- It has been iterated that leiomyosarcoma of the para-testicular region is the second most common malignant mesenchymal tumour of the para-testis, after liposarcoma of the para-testicular region. [4]
- It has been pointed out that 10% to 30% of para-testicular sarcomas are leiomyosarcomas [4] [5] [6]
- It has been documented that the 5-year and 10-year disease-specific survival rates of leiomyosarcoma of the para-testicular region are 77% and 66%, respectively [4]
- It has been documented that after a 4-year follow-up assessment, patients who had been afflicted by leiomyosarcoma of the para-testicular region, 30% had metastases to the lymph nodes, lungs, and liver in all grade 3 tumours [7]
- Median age is 64 years, range 17 - 92 years [7] [8]
Sites
- It has been documented that the spermatic cord is the most common para-testicular site of leiomyosarcoma [7]
- Other sites of leiomyosarcoma within the scrotum had been documented to include: the epididymis and scrotal sac [9]
Pathophysiology
The pathophysiology of leiomyosarcoma of the para-testicular region has been summarised as follows: [3]
- It has been iterated that leiomyosarcoma of the para-testicular region has been hypothesised to arise from cremasteric muscle, vas deferens or contractile cells of other structures such as tunica or blood vessels [8]
- It has been iterated that para-testicular leiomyosarcoma tumours belong to the "deep type" of leiomyosarcoma
- It has been pointed out that no recurrent genetic abnormality has been documented in primary leiomyosarcoma of the para-testicular region.
- It has been iterated that the leiomyosarcoma of the para-testicular region tumour spreads via lymphatic and hematogenous metastases or by local extension of the tumour.
- It has been iterated that lymphatic spread of leiomyosarcoma of the para-testicular region is to the regional lymph nodes: external iliac, hypogastric, common iliac, retroperitoneal [10]
- It has been documented that the lung is the most common site for hematogenous spread of primary leiomyosarcoma of the para-testicular region.
Clinical features
The clinical features of primary leiomyosarcomas of the para-testicular region had been summarised as follows: [3]
- Typically, primary leiomyosarcomas of the para-testicular region do manifest with a painless, slow-growing mass (at least a few months) within the scrotal sac
- Rarely, primary leiomyosarcoma of the para-testicular region organ presents with a sudden increase in size of a long-standing intra-scrotal mass
- Aggressive tumours may invade the dartos muscle and overlying skin, causing a fungating mass
Laboratory
- It has been iterated that in primary leiomyosarcoma of the para-testicular region, there tends to be normal serum tumour markers (LDH, β-HCG, α-fetoprotein), which helps to exclude a germ cell tumour of the testis [3]
Radiology description
The radiology-image features of primary leiomyosarcoma of the para-testicular region had been summated as follows: [3]
- Ultrasound scan of the scrotum and scrotal contents demonstrates a mass with mixed echogenicity and increased vascularity [11]
- Colour Doppler ultrasound scan of the scrotum and scrotal contents in a case of primary leiomyosarcoma of a para-testicular region organ demonstrates an irregular vascularity, which is more dominant within the periphery, similar to malignant tumours
- CT scan usually demonstrates absence of fat (absence of foci with negative Hounsfield units).
- A variable degree of hydrocele may be present
Prognostic factors
The factors of prognostication of primary leiomyosarcoma of the para-testicular region organs had been summated as follows: [3]
- The prognostic factors are similar to the prognostic factors of sarcoma, which include the following: the tumour size, the tumour grade, presence of necrosis, mitotic count, completion of resection, local recurrence, and distant metastases
- Specific prognostic criteria of primary leiomyosarcoma of the para-testicular-region organs are not well established due to the rarity of the tumour
- FNCLCC grading, which has been recommended over the NIH system, gives points for the following:
- Differentiation: 1, well-differentiated leiomyosarcoma; 2, conventional leiomyosarcoma; 3, poorly differentiated / pleomorphic/epithelioid leiomyosarcoma
- Mitotic count: 1, 0 - 9 per 10 high-powered fields; 2, 10 - 19 per 10 high-powered fields; 3, 20 or more per 10 high-powered fields
- Necrosis: 0, none; 1, less than or equal to 50%; 2, greater than 50%
- Grade 1 is 2 - 3 points, grade 2 is 4 - 5 points, and grade 3 is 6 - 8 points
Treatment
The treatment of leiomyosarcoma of the para-testicular region had been summarised as follows: [3]
- Radical orchidectomy tends to be undertaken for resectable primary leiomyosarcoma tumours of the para-testicular region
- Simple excision of para-testicular leiomyosarcoma is stated to be insufficient as residual disease was found in 27% of cases that underwent repeat wide excision [12]
- It has been pointed out that the role of adjuvant radiotherapy or chemotherapy in the treatment of primary leiomyosarcoma of the para-testicular region is not certain and that some authors recommend adjuvant therapy in high grade / high risk tumours and others recommend adjuvant radiotherapy for all grades to reduce local recurrence rate of the tumour [13] [14]
- It has been iterated that the undertaking of prophylactic retroperitoneal lymph node dissection in cases of primary leiomyosarcomas of the para-testicular region may be undertaken, but no survival benefit has been documented, so dissection had been recommended only if the lymph nodes are suspicious for involvement of the tumour [10]
- About primary leiomyosarcomas of the para-testicular region organs, it has been iterated that single or multi-agent chemotherapy with anthracyclines may be provided and if osfamides or gemcitabine for non-operable or for metastatic tumours [3]
Gross description
The macroscopy pathology examination features of specimens of primary leiomyosarcomas of the para-testicular region had been summated as follows: [3]
- The tumour typically is typically found to be located outside the tunica albuginea, and the tumour may be centred upon the epididymis or spermatic cord
- The cut surface of specimens of primary leiomyosarcomas is usually firm and grey-white
- Grossly identifiable necrotic and haemorrhagic areas may be visualised
Microscopic (histologic) description
The microscopy pathology examination features of specimens of primary leiomyosarcomas of the para-testicular region organs had been summated as follows: [3]
- Interlacing fascicles of spindle-shaped cells with eosinophilic cytoplasm and cigar-shaped nuclei are visualised upon microscopy and histopathology examination of the specimen
- Mitotic activity within the tumour is usually seen, and necrosis may be visualised
- Microscopy examination of the tumour specimen, at least, tends to demonstrate mild atypia, and more undifferentiated cases do contain features of marked cellular atypia
- Variably scattered mast cells, inflammatory cells, and hyalinization tend to be visualised upon microscopy examination of specimens of leiomyosarcoma of the para-testicular region organs.
- Histological variants of primary leiomyosarcoma of the para-testicular region include the following:
- Epithelioid
- Dedifferentiated/pleomorphic
- Discrete transition to more pleomorphic tumour (reminiscent of malignant fibrous histiocytoma) without any smooth muscle differentiation
- Dedifferentiated areas are usually negative for all myogenic markers
- Heterologous osseous or chondro-osseous elements may be seen rarely
- With osteoclast-like giant cells [15]
- Myxoid [16]
- Inflammatory
Immunohistochemistry & special stains
- Positive:
- Muscle specific actin; smooth muscle actin; h-caldesmon; and desmin
- Vimentin
- Variable:
- CD34
- Negative / focal rare cells:
- Cytokeratin, CD117, myogenin, myoD1, S100
Electron microscopy description
- 7 nm microfilaments, regular dense bodies, micropinocytotic vesicles, glycogen pools [17]
Differential diagnoses
The differential diagnoses of primary leiomyosarcoma of the para-testicular region organs had been summed up as follows:
- Dedifferentiated liposarcoma with smooth muscle differentiation
- More typical liposarcomatous areas
- Fibromatosis: tapered nuclei
- Leiomyoma
- No appreciable mitotic activity or necrosis, but nuclear atypia may be present
- Scrotal smooth muscle tumours should be classified as potentially malignant if any mitotic activity is seen [20]
- Malignant peripheral nerve sheath tumour
- Mesothelioma
- Associated with hydrocele, asbestos exposure; infiltrative margins; usually keratin+, desmin-
- Solitary fibrous tumour
- Disorganised spindle cells, diffusely CD34+ and desmin-
- Spindle cell rhabdomyosarcoma
- Usually occurs in children Myogenin
[B] Miscellaneous Narrations and Discussions from Some Case Reports, Case Series, And Studies Related to Leiomyosarcoma of the Urinary Bladder
Saidani et al. [18] undertook a study to expose the experience of their department in dealing with urinary bladder leiomyosarcomas and illustrate the management tools of this rare pathology. Saidani et al. [18] undertook a retrospective study of 4 cases of urinary bladder leiomyosarcoma, which were gathered within the Department of Urology during the period of 1996–2022. All of the patients were exclusively male and who were aged between 35years and 73 years. No history of pelvic radiotherapy nor chemotherapy was found in the patients. Three patients had high-grade leiomyosarcoma and pT2 stage whereas only one had a low-grade tumour. Three patients underwent radical treatment by cystoprostatectomy with extensive bilateral pelvic lymph node dissection and one patient was treated by endoscopic re-resection and endoscopic monitoring. Saidani et al. [18] noticed 2 lymph nodes and liver metastasis recurrences in 2 patients who were treated by radical surgery while 2 patients did not present recurrences at two years of follow-up. Saidani et al. [18] made the ensuing discussions:
- Up to 2023, there had not been any clear and precise treatment approach for the treatment of urinary bladder leiomyosarcoma.
- Little is known about the long-term survival associated with these tumours.
- All reported studies had agreed that the prognosis for urinary bladder leiomyosarcoma is poor, if not diagnosed early, especially those manifesting with an undifferentiated tumour grade, distant metastasis and treated without surgical therapy.
Saidani et al. [18] made the ensuing conclusions:
- Urinary bladder leiomyosarcoma is a rare and highly aggressive tumour.
- Pathology examination provides the diagnosis and prognosis assessment.
- The undertaking of radical surgery remains the most suitable treatment option.
Hamadalla et al. [19] reviewed the published cases of leiomyosarcoma of the urinary bladder and reported two further cases. Hamadalla et al. [19] searched the databases including Pubmed and Hinari using the keywords ‘bladder’, ‘leiomyosarcoma’ and ‘smooth muscle neoplasm’. They reviewed the 14 articles they had identified and they presented a further two cases. Hamadalla et al. [19] summated the results as follows:
- Of more than 100 cases that had been reported, 77 were reviewed.
- There is a lack of consensus about the standard treatment, and little is known about the natural history and prognosis of the tumour, because of its very low incidence.
- These tumours afflict older adults of either sex and are characterised by an aggressive behaviour.
- There is usually an unfavourable outcome, with the lungs being the most common site of metastasis. T
- he two further cases they had reported had a different presentation and outcome.
Hamadalla et al. [19] concluded that:
- Because of the limited experience with this rare tumour, there are insufficient data to indicate the optimum management strategy and prognosis.
Zieschang et al. [20] made the ensuing iterations:
- Leiomyosarcoma of the urinary bladder is exceedingly rare.
- Most clinicians come across only a few cases during their career, and information regarding treatment and outcome is scattered in the scientific literature.
- Interested clinicians and patients need to undertake a troublesome search for treatment and outcome information.
Zieschang et al. [20] undertook a systematic review of the literature utilising the PubMed and Web of Science databases that included all identified cases published in the English language between 1970 and June 2018 into a meta-analysis. Before the literature search, key questions were formulated, and with the data obtained, answers to these questions should be derived. Zieschang et al. [20] summated the results as follows:
- They had analysed clinical data of 210 cases of urinary bladder leiomyosarcoma, which were revealed by their review and seen in their institution.
- The mean age of the patients was 52 years.
- The majority of the tumours, which accounted for 75% of the tumours was classified as high-grade sarcomas.
- They found no report of a prior radiotherapy to the pelvic organs, but some authors had suggested an association between cyclophosphamide treatment and the development of urinary bladder leiomyosarcoma, especially in patients with retinoblastoma.
- For the whole sample, they had determined the 5-year and 10-year cancer-specific cumulative mortality rates of 38% and 50%.
- Patients who had high-grade sarcomas had a trend toward a higher mortality compared with low-grade tumours (p = 0.0280).
- The most promising treatment option seemed to be surgery (radical or partial cystectomy) with negative resection margins, possibly supplemented by chemotherapy or radiation.
Zieschang et al. [20] concluded that:
- About half of patients with urinary bladder leiomyosarcoma survived in the long run.
- Low-grade tumours may have a better outcome, with, however, countable long-term mortality.
- For better assessment of that rare bladder tumour, its best treatment options, and the influence of neoadjuvant or adjuvant therapies on the outcome of patients, a larger series with long-term survival data is necessary.
Tanquay et al. [21] stated that: Leiomyosarcoma of the urinary bladder is rare, even though it is the most common mesenchymal tumour in adults. Tanquay et al. [21] reported two cases of this tumour following cyclophosphamide therapy. The first case was from a 53-year-old man with Wegener's granulomatosis who was treated for 6 years with cyclophosphamide. He manifested with painless visible haematuria, and the initial biopsy of his urinary bladder tumour upon pathology examination demonstrated a malignant spindle cell neoplasm. A final diagnosis of leiomyosarcoma was made on radical cystoprostatectomy. The second example was from a 21-year-old man who had received cyclophosphamide in his early infancy for a bilateral retinoblastoma. He also manifested with painless visible haematuria, and a bladder tumour was resected transurethrally and pathology examination of the bladder tumour specimen confirmed a diagnosis of leiomyosarcoma of the urinary bladder. He underwent partial cystectomy two months later. Tanquay et al. [21] made the ensuing iterations:
- Cyclophosphamide, when used for a neoplastic or non-neoplastic condition, is associated with an increased risk of developing urinary bladder cancer.
- The distribution of histology subtypes differs from that seen in spontaneous urinary bladder tumours.
- A review of the literature has shown an increased proportion of squamous cell carcinomas and sarcomas, especially leiomyosarcomas, in cyclophosphamide exposed patients.
- Acrolein, which is a cytotoxic metabolite of cyclophosphamide excreted in urine, is regarded as the most likely causative agent.
Tanquay et al. [21] made the ensuing iterations:
- Non-epithelial tumours of the urinary bladder account for less than 5% of the overall bladder malignancies, with leiomyosarcoma being the 0.1% of bladder cancer [22].
- By 2010, there were over 100 cases reported in the whole medical literature as leiomyosarcomas, and a total of 192 cases considering all bladder sarcoma cases [23].
- There is a lack of consensus about a standard treatment, and little is known about the natural history and prognosis of the tumour, due to a very low incidence.
- Cases which had been presented in the literature had mostly gross urinary symptoms, more often with an earlier onset. This may, eventually, lead to earlier diagnosis, enabling a safer treatment approach.
- The case they had reported showed an unusual manifestation, differing from the symptoms reported in the literature, simulating mostly a pelvic mass. The patient underwent, in the early diagnostic steps, a gynaecologic evaluation which led to a final multidisciplinary surgical approach to the disease. The high-grade and stage of the tumour, in association with the fatal outcome of the case, demonstrate the expression of the aggressive behaviour of the tumour, showing mostly a very poor prognosis.
Ricciardi et al. [2] made the ensuing iterations:
- Mesenchymal tumours represent a small number of bladder cancer cases.
- Leiomyosarcoma is the most common histology, with over 100 cases reported in the literature.
- This tumour had been historically considered to be highly aggressive and demonstrated a poor prognosis.
- Despite very low survival rates shown in older reports, some authors had indicated that some patients could have a better outcome.
Ricciardi et al. [2] reported a review of the literature and a case of high-grade LMS of the bladder in a 68-year-old woman. Diagnosis was delayed, and the disease was locally advanced. The symptoms and radiology-imaging of the reported case first suggested a gynaecologic condition with an adnexal or uterine origin of the mass, and a genitourinary origin could be unveiled only intra-operatively. Ricciardi et al. [2] reported a case of a 68-year-old woman who had presented to the emergency department with visible haematuria and dysuria. She had experienced fever several times, commencing two months before her medical examination. Her fever was treated with NSAID. A history of recent pelvic pain was also reported. Her clinical status, as determined by her assessment determined to be good and no other symptoms or concurrent illnesses were present at the time of hospitalisation. She was admitted to the Gynaecologic service for further evaluation. The patient was a virgin and in post-menopausal status. Her clinical history was unremarkable; negative either for previous medical or surgical procedures. Her family history was positive for cancer with one male sibling who was deceased from a pancreatic malignancy; the remainder two siblings had no positive history for malignancies as well as parents and closest relatives. Her social history was negative for use or addiction to drugs, alcohol and exposure to potential risk factors. A digital rectal examination had to be undertaken in place of the gynaecologic bimanual examination. A gross, firm mass located within her pelvis was found during her physical examination. She had pelvic ultrasound (US), abdominal CT scans, descending pyelography, cystoscopy and chest radiographs were undertaken to both assess local the disease and evaluate for the presence of local and distant metastases. A computed tomography imaging was undertaken, which confirmed the presence of a complex mass in her pelvis that measured 13 cm × 14 cm, demonstrating irregular contours and small areas of calcifications. Her uterus and adnexa could not be clearly recognized, showing as being part of the mass. Her Iliac vessels and bladder were displaced. The radiology-imaging technique report stated that no vessels or urinary bladder invasion was present. A hypoplasia of the left kidney was also reported. No other metastasis was reported within the abdomen and pelvis.
She had a pyelography which demonstrated a delay in the opacification of the renal pelvis associated to a dilation of the right ureter that was compressed and displaced in its pelvic course till entering the bladder. No opacification of the contralateral kidney and excretory ways could be obtained. Cystoscopy was undertaken, which showed, eventually, a bleeding lesion on the urinary bladder wall that raised suspicion of neoplastic invasion. The lesion appeared more likely as a loss of continuity of the urothelial mucosa; an ulcerating mass was visualised and eventually described on the final report. She had a chest RX and a bone scan, which were negative for distant metastases. A multidisciplinary board, including a urologist, a radiologist, a radiation oncologist, an oncologist and a gynaecologist, had evaluated the collected data. It was eventually decided to surgically extirpate the tumour. A transurethral biopsy of the vesical wall lesion was not considered since the major suspicion was addressed to uterine or adnexal neoplasms, and the high stage was reasonably an indication to undergo definitive surgery. Surgery was eventually undertaken after the pre-operative routine and evaluation. A longitudinal abdominal incision was preferred to access the peritoneal cavity. A bulky mass was found within her pelvis. Her uterus and adnexa appeared macroscopically free from neoplastic invasion. The mass demonstrated a cleavage with the recto-sigmoid tract, iliac vessels and pubic bone, whether uterus, adnexa, right ureter and left obturator nerve could not be separated from the tumour. A histological intra-operative examination report revealed a malignant, spindled cell neoplasm. Her surgery had to include a wide mass excision, to obtain free margins and control of local disease. The final treatment choice was a pelvic exenteration. This decision was made since her local disease was advanced and it was set to obtain specimen's margins disease-free as a primary target. An en bloc resection of the mass, together with the uterus, adnexa, bladder, distal part of right ureter and left obturator nerve was undertaken, which amounted to anterior pelvic exenteration. A cutaneous ureterostomy was inserted in the site of ureteral dissection. The decision to undertake this procedure in place of a standard urinary diversion, as an ileal conduit or Miami pouch, was taken based upon the evidence of poor prognosis that appeared from surgical exploration and intra-operative histology that addressed a malignant stromal neoplasm. It seemed to be reasonable to lower intra- and post-operative morbidity, whereas the potential overall survival of the patient appeared to be prospectively low.
A final histopathology diagnosis of high-grade leiomyosarcoma, (see figure 1), (G3, FNCLCC 1986) pT2b pN0 pM0, AJCC (2002) Stage III, was made based upon immunoreactivity to smooth muscle actin, score 2 (see figure 2). The tumour specimen exhibited negative staining for c-kit and EGFR. Her proliferation index was 90%, which was evaluated thru MIB-1 (Ki-67) (see figure 3). Necrosis was inferior to 50% (score 2) and mitotic index higher than 20 mitoses per field/10 HPF (score 3). The tumour presented, macroscopically, as a bulky, brain-like, white-greyish mass. The tumour was noted to contain diverse necrotic and haemorrhagic areas, and invaded the vesical wall. The size of the mass was 14 cm × 11 cm × 6.5 cm. Histopathology examination of the specimen confirmed that the uterus and adnexa were free from neoplastic invasion. The surgical margins resulted negative at the final examination. LVI was not reported.
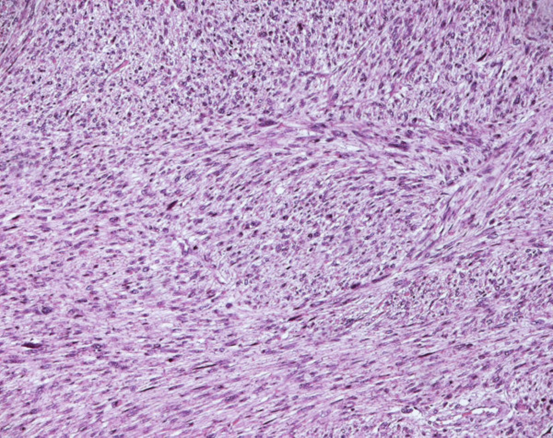
Figure 1.
Leiomyosarcoma of the bladder (Hematoxylin and eosin). Reproduced from: [2] under Creative Commons Attribution License
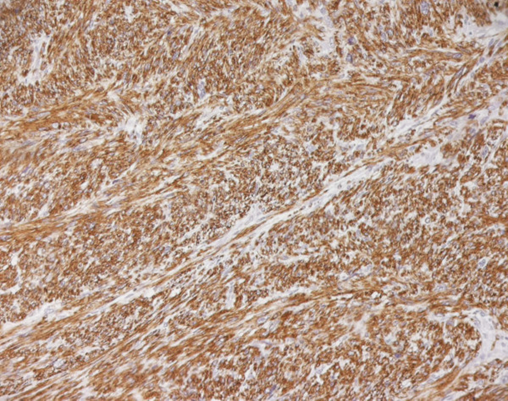
Figure 2.
Leiomyosarcoma of the bladder (Smooth muscle actin). Reproduced from: [2] under Creative Commons Attribution License
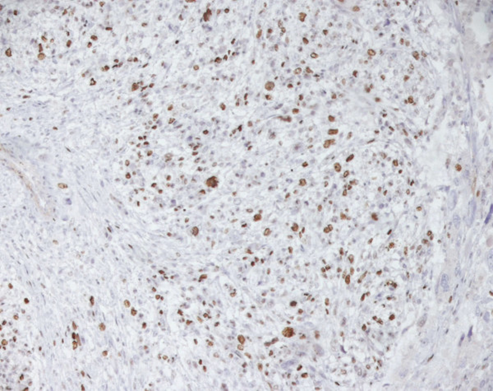
Figure 3.
Leiomyosarcoma of the bladder (Ki67). Reproduced from: [2] under Creative Commons Attribution License
The post-operative course was free from complications, and the patient could be discharged from the hospital fourteen days after surgery. Ep non-epithelial tumours of the urinary bladder account for less than five per cent (5%) of overall malignancies of the urinary bladder, with leiomyosarcoma constituting 0.1% of urinary bladder cancers the patient underwent chemotherapy one month after her surgery, using doxorubicin as a single agent in an adjuvant setting. She died after the first cycle from a distant recurrence in the left lung.
Ricciardi et al. [2] made the ensuing discussions:
- Sarcoma is the most frequent mesenchymal malignancy of the urinary bladder, with leiomyosarcoma as the most common histology, according to two retrospective reviews of mesenchymal genitourinary tumours. [24] [25]
- Considering the whole of the cases that had been reported in the literature (192 in total), 50% were leiomyosarcomas, 20% rhabdomyosarcomas and the remainder consisting of other histologies as carcinosarcomas, angiosarcomas and osteosarcomas. [23]
- No specific risk factors had been identified yet for this tumour.
- Reviewing the literature, retinoblastoma (RB) gene mutations were identified among possible causes in at least 9 cases, as well as the utilisation of cyclophosphamide. [26] [27] [28] [29]
- Pelvic radiotherapy for other malignancies was also described in literature as a risk factor present in the clinical history of the patient.
- According to Rosser et al., the most common clinical presentation is visible haematuria (81%), dysuria (19%) and pollakiuria (28%).
- Their reported case had presented mostly as a pelvic mass, showing urinary symptoms similar to those described by Rosser et al. [30] but very late in the disease course.
- The diagnosis in their case was therefore delayed, due on the fact that the patient had no symptoms until the tumour had reached an advanced stage and became locally invasive. The patient, otherwise being well, did not consider undergoing a medical evaluation, until the onset of urinary symptoms.
- Initial reports for this tumour had shown a very aggressive disease.
- In a series by Mackenzie et al. which was published in 1968, only 11 patients were alive at 3 years pursuant to surgery. [31]
- Further series had shown that the outcome which was shown in previous series, could be better that once believed.
- A series at MD Anderson considered 19 patients with a bladder sarcoma diagnosis. It showed a 5-year disease-specific survival of 59%. They also reported lymph-vascular invasion and node status as significant prognostic markers, claiming a 3.4 median survival for patients with no LVI in front of a 0.8 median survival when LVI was present. Pathologic node status showed a median survival of 7.6 years when negative and 0.3 years when positive. [32]
- The largest series, by Rosser et al. [30] which included 35 patients, with a diagnosis of LMS of the bladder, showed a 5 years disease-specific survival of 62%, with a 34% recurrence rate at a median follow-up of 38 months, with both local and distant disease sites affected. [30]
- Nevertheless, the best prognostic factor seemed to be the presence of free margins.
- In their case, they had achieved disease-free margins; still, they did not determine a better outcome. In addition to margin status, local invasiveness and size, as well as tumour grade, seem to play an important role in determining the outcome. In their case, as stated above, LVI was unavailable.
- According to one of the greatest series available to date, overall local recurrence of bladder leiomyosarcomas is about 16%, with most recurrences occurring in the pelvis. Overall recurrence of distant metastases is about 53%, with the most common sites of metastases being the lungs, liver, bone, and brain. [32]
- Because of the very low incidence, there is no universal consensus opinion on the treatment of patients affected.
- Minimally invasive approaches, such as transurethral resection or laser fulguration, in addition to CTX or RT adjuvant settings, have been used for patients with small lesions.
- Long-term survival rates did not differ significantly from cases which had undergone a more radical surgery.
- A sole resection of the mass, even when free margins were achieved, as in the case of partial cystectomy, should be considered a palliative treatment. [33] [34] [35]
- The main treatment consists of radical cystectomy (including removal of uterus, cervix and vaginal cuff in women). The procedure should include wide margins resection with 2-3 cm depth free from tumour invasion. [36] [37] [38] [40]
- Neoadjuvant and adjuvant therapies were utilised in 21% and 16% of patients at MD Anderson, respectively, and both resulted in a doubling of disease-specific survival. Nevertheless, this result was found not to be statistically significant, reflecting the small numbers of patients in each group. Similarly, it is difficult to evaluate the impact of neoadjuvant and adjuvant chemotherapy upon quality of life. [32]
- A multimodal treatment with CTX settings targeting mesenchymal cancers (as sarcoma chemotherapy protocols using doxorubicin, ifosfamide, cisplatinum and docetaxel) should be mandatory in the event of metastatic disease. [32]
- With patients who have positive margins after surgery, adjuvant radiotherapy should be advocated for the patient. [39]
- Local recurrences should be treated by systemic CTX and/or external pelvic RT, salvage therapy has been shown to be ineffective, with a median survival of 20 months after surgery. [32]
Ricciardi et al. [2] made the ensuing conclusions
- Sarcomas should be considered as a possible histology in differential diagnosis, even if they are not as frequent compared to other tumours.
- Unfortunately, obtaining a pre-operative histopathology diagnosis does not improve the prognosis.
- Rare tumours represent a great challenge for physicians. They require experienced teams and well-equipped centres for cancer cure.
- Urinary bladder sarcomas, as other genito-urinary sarcomas, require close cooperation between urologist and gynaecologic oncologist, as well as medical and radiation oncologist.
- A pathologist expert in mesenchymal tumours is mandatory.
- Nowadays, no statistically relevant evidence on treatment behaviour can be found in the literature.
- Therefore, the treatment should be tailored case-by-case, preferring a multimodal and/or multidisciplinary approach to the disease.
- A decision-making team made by physicians who are experienced in managing soft tissues sarcomas should be also strictly required.
Fakhri et al. [41] made the ensuing iterations:
- Leiomyosarcomas of the urinary bladder are rare smooth muscle tumours, and constitute 1% of all bladder malignancies.
- Even though rare, leiomyosarcoma is a clinically significant disease, presenting as a high-grade advanced malignancy with associated substantial morbidity and mortality, if not treated early.
- The diagnosis could be delayed because of the asymptomatic presentation until the tumour reaches an advanced stage.
Fakhri et al. [41] presented two cases of leiomyosarcoma of the urinary bladder. The first case: a 63 years old female with a history of cervical carcinoma treated with radiation, 25 years earlier, followed by uncomplicated clinical course, until her recent development of vesicovaginal fistula. During her workup with cystoscopy, a large bladder mass was identified. Second case: a 71-year-old female with a history of treated breast cancer, who had presented with a recurrent history of cystitis and haematuria. Cystoscopy had demonstrated a nodular mass in the right posterior bladder wall. Fakhri et al. [41] summated the pathology results as follows:
- Microscopically, both lesions were found to be highly cellular with infiltrative, interlacing fascicles of spindle cells and associated mitosis, cellular atypia and necrosis.
- Immunohistochemistry studies had revealed positive expression of muscle- specific-actin, desmin, and caldesmon; with negative expression of ALK-1, EMA, Cytokeratins and GATA3, in both cases.
- Other spindle cell lesions, including leiomyoma, inflammatory myofibroblastic tumour and sarcomatoid carcinoma, were included in the differential diagnoses, and they were excluded by morphology and immunohistochemistry.
Fakhri et al. [41] made the ensuing conclusions:
- Their cases had highlighted the importance of awareness of the occurrence of leiomyosarcomas, a rare manifestation in bladder.
- Even though rare, they present with a poor outcome.
- The differential diagnoses of spindle cell lesions is extensive, including close benign and malignant mimics, the treatment of all being very different. Immediate radical cystectomy has been associated with longer survival rates for leiomyosarcoma.
- Leiomyosarcoma, being aggressive, with an associated high recurrence rate and metastatic potential, an early correct diagnosis would aid in the institution of proper management.
Parekh et al. [23] reported an unusual case of a leiomyosarcoma of the urinary bladder after chemotherapy for retinoblastoma and the results of a review of the published reports of bladder sarcomas. Parekh et al. [23] reported a 22-year-old man who had manifested with visible haematuria and who was found to have a mass within his urinary bladder on computed tomography. Transurethral resection of the mass was undertaken and pathology examination of the specimen revealed a leiomyosarcoma. The patient underwent radical cystectomy and an ileal conduit was created. The final pathology examination confirmed high-grade leiomyosarcoma. Parekh et al. [23] concluded that:
- Prospective randomized combination trials similar to the Intergroup Rhabdomyosarcoma Study in the paediatric population are necessary to better understand and manage these potentially curable sarcomas.
Brucker et al. [42] described a twin with bilateral retinoblastoma who developed leiomyosarcoma of the bladder at age 17 years and again at 39 years. At 17-years of age she was diagnosed with a leiomyosarcoma of the urinary bladder after manifesting with recurrent urinary tract infections, haematuria, and dysuria. She was treated with partial cystectomy. After a 12-year disease-free interval, she was diagnosed with a second leiomyosarcoma of the bladder. Brucker et al. [42] concluded that:
- Their reported case had supported the relationship between the genetic form of retinoblastoma and leiomyosarcoma and had illustrated the necessity for extensive follow-up and well-defined treatment of secondary neoplasms.
Al-Zahrani et al. [43] reported a 16-year-old female with urinary bladder leiomyosarcoma who had had a history of bilateral retinoblastoma at 6 months of life. She received cyclophosphamide chemotherapy after surgical enucleation.
Motta et al. [44] reported a case of urinary bladder leiomyosarcoma which occurred in a 22-year-old female who had been treated with cyclophosphamide for a period of 68 months for retinoblastoma, which was diagnosed at 18 months postpartum. Partial cystectomy was undertaken. Forty-two months after the operation, she had been tumour-free.
Kawamura et al. [45] reported a a case of leiomyosarcoma within the urinary bladder which occurred in an 18-year-old boy who had been treated with cyclophosphamide over a period of 6 years for retinoblastoma which was diagnosed 50 days postpartum. Total cystectomy and formation of an ileal conduit were undertaken. Three years after the operation, he was tumour-free.
Ramírez Sevilla et al. [46] presented a rare urinary bladder tumour in a young patient 25 years after the treatment with cyclophosphamide because of a neuroblastoma of the right eye. The first symptom of the tumour was visible haematuria with dysuria and pollakiuria. The final diagnosis was determined by the pathologist and the best treatment option was radical cystoprostatectomy. Leiomyosarcoma was presented in a 26-year-old patient, the third different tumour, the second was an esphenoidal meningioma. At the 8th year of follow-up after the radical surgery, the patient was free of bladder disease. Ramírez Sevilla made the ensuing conclusion:
- Urinary bladder leiomyosarcoma is a rare tumour of the bladder, and its early diagnosis and treatment are mandatory for the best prognosis.
Minagawa et al. [47] reported a case of urinary bladder leiomyosarcoma in a 27-year-old woman who had previously been treated with surgery and radiotherapy for bilateral retinoblastoma. The patient was admitted to the hospital with discomfort on micturition. She underwent cystoscopy, which demonstrated a urinary bladder tumour which was covered by normal urothelium. Transurethral resection of the bladder tumour was undertaken, and the histopathology diagnosis was leiomyosarcoma. Partial cystectomy was undertaken. The leiomyosarcoma of the bladder did not invade the muscle layer. Nevertheless, the bladder tumours recurred at new intravesical locations repeatedly. After transurethral resection of the bladder tumour had been undertaken twice, total cystectomy and creation of an ileal conduit were undertaken subsequently.
Kin et al. [48] et al reported a 41-year-old woman, who was admitted with visible haematuria and pain on urination. She had cystoscopy which demonstrated a huge and lobulated submucosal non-papillary bladder tumour. She had pelvic computed tomography which demonstrated a heterogeneous and enhanced lobulated mass, 8 cm in diameter, with extravesical invasion but there appeared to be no metastatic lesions. Transurethral biopsy of the specimen revealed leiomyosarcoma pathologically. Total cystectomy and construction of an ileal conduit were undertaken. The tumour was histologically diagnosed as leiomyosarcoma. Immunohistochemical studies of the tumour revealed the tumour cells had exhibited positive staining for epithelial membrane antigen (EMA) and muscle actin but negative staining for desmin, and S-100. Kin et al. [48] reviewed 102 cases of vesical leiomyosarcoma reported in Japan. Among these 102 cases, there were no EMA-positive cases. Kin et al. [48] concluded that:
- Immunohistochemical and electron microscopy evaluation should be performed to evaluate this disease.
Labaris et al. [49] made the ensuing iterations:
- Non-urothelial neoplasms of the bladder account for fewer than 5% of all bladder tumours.
- Sarcoma constitutes the most common mesenchymal malignancy of the bladder, with leiomyosarcomas being the most common type of sarcoma in adults.
Labaris et al. [49] examined the records of seven patients presenting to two different institutions with bladder leiomyosarcomas between 2003 and 2007 and between 2000 and 2007, respectively. Cystoscopy, with transurethral resection of the bladder tumour, was initially undertaken in all patients, with leiomyosarcoma being initially diagnosed based on examination of the transurethral specimen. Labaris et al. [49] summated the results as follows:
- There were N = 5 men and N = 2 women with a median age of 64.8 years at presentation.
- All seven patients had undergone a definitive surgical procedure.
- Complete resection with negative surgical margins was achieved in all seven patients (100%).
- MSKCC stage had included 86% of patients with stage 3 (N = 6) and 14% with stage 2 (N = 1).
- A low-grade tumour was evident in one patient only, with the remaining 86% exhibiting a high-grade tumour.
Labaris et al. [49] made the ensuing discussions:
- Leiomyosarcomas of the urinary bladder had always been considered a highly aggressive entity, and little is known about their origin, clinicopathologic presentation, and the survival factors associated with them.
- Contemporary studies have suggested that these tumours might have a better prognosis than was once believed.
- Leiomyosarcomas require aggressive surgical extirpation and, when surgical resection is possible, radical cystectomy with wide margins is the rule and should be undertaken.
- Strict adherence to standard surgical technique had resulted in low rates of positive surgical margins and low rates of local tumour recurrence.
Lindberg et al. [50] made the ensuing iterations:
- Leiomyosarcomas of the urinary bladder (LMS-UB) are rare, usually aggressive neoplasms.
- Owing to their rarity, only a limited number of cases of LMS-UB with clinical follow-up information had been published.
- There is no current consensus opinion on LMS-UB grading, and it is unknown whether the widely accepted Fédération Nationale des Centres de Lutte Contre le Cancer (FNCLCC) and National Cancer Institute (NCI) grading systems of soft-tissue sarcomas apply to LMS-UB.
Lindberg et al. [50] studied 34 well-characterised LMS-UB and compared the prognostic power of the FNCLCC and NCI systems with that of one published grading scheme for LMS-UB (Mayo). Lindberg et al. [50] retrieved and evaluated all available slides from 34 LMS-UB about the degree of differentiation, mitotic rate/10 high-powered fields (HPF), and % necrosis. Lindberg et al. [50] graded the cases utilising published criteria for the FNCLCC, NCI and Mayo schemes. Follow-up information was obtained. Lindberg et al. [50] summated the results as follows:
- The tumours had afflicted 17 females and 17 males, whose ages had ranged from 31 years to 91 years, and their median age was 65 years, as well as the tumours measured between 2 cm and 12 cm in size.
- One tumour was observed to be well differentiated, 17 tumours were moderately differentiated, and 16 tumours were poorly differentiated.
- The mitotic rates had ranged from 1 to >30/10 HPF (median 12/10 HPF), and tumours showed 0-60% necrosis (median 25%).
- The FNCLCC grades of the tumours were 1 in 3 cases, 2 in 12 cases and 3 in 19 cases.
- The NCI grades were 1 in 2 cases, 2 in 11 cases and 3 in 21 cases.
- The Mayo grades were low in 7 cases and high in 27 cases.
- FNCLCC and NCI grades were identical in 23 out of 34 cases, which amounted to in 68% of the cases.
- Four cases were FNCLCC/NCI grade 2 or 3 and Mayo low-grade.
- Clinical follow-up was available for 25 of 34 patients which amounted to in 74% of the cases.
- Clinical follow-up of longer than or equal to twelve (> or =12) months was available for 17 of these 25 cases which amounted to 68% with a median follow-up duration of 52 months and the follow-up had ranged between 12 months and 120 months.
- Adverse outcome was seen in nine of these 17 patients which amounted to in 53% of the patients.
- Seven out of the eight cases, which amounted to 88% of the cases with a clinical follow-up duration of less than (<) 12 months died of their disease.
- Overall, an adverse outcome was documented in 16 of 25 (64%) cases. Metastatic disease was seen in 13 of 25 (52%) cases, with the lungs being the most common site of metastasis (62%). Adverse outcome was noted in 15 of 23 patients which amounted to in 65% of patients who had FNCLCC grade 2 or 3 LMS-UB, as compared with zero of two (0%) FNCLCC grade 1 tumours (p=0.15), in 15 of 23 cases which amounted to 65?ses with NCI grade 2 or 3 LMS-UB, versus zero of two (0%) NCI grade 1 sarcomas (p=0.17) and in 13 of 20 (65%) Mayo high grade LMS-UB, as opposed to two of five (40%) low-grade lesions (all results not statistically significant).
Lindberg et al. [50] concluded that:
- LMS-UB occurs in older adults of either sex and is typified by aggressive behaviour, with adverse outcome in greater than (>) 60% of cases.
- Certain advantages of the FNCLCC system might support its more widespread adoption for future studies.
Benjamin et al. [51] made the ensuing iterations:
- No well-established staging system exists for urinary bladder leiomyosarcoma (LMS), and the current staging system does not include tumour size, a thoroughly validated prognostic parameter for sarcomas.
- Uterine and extremity/trunk LMS are more common than those in the urinary bladder and have well-established staging systems incorporating the tumour size.
- They had aimed to improve the understanding of LMS of the urinary bladder by assessing cancer-specific survival (CSS) and comparing LMS at this unusual anatomic site to those arising at other sites using the Surveillance,
Benjamin et al. [51] queried the Epidemiology and End Results (SEER) database. The SEER database (1973–2013) for bladder, uterus, and trunk/extremity LMS. Benjamin et al. [51] undertook Multivariable Cox proportional hazard regression to identify predictors of CSS for each anatomic location and used it to compare outcomes at different sites. Benjamin et al. [51] summated the results as follows:
- They had identified 165 urinary bladder, 4987 uterus, and 2536 extremity/trunk LMS cases.
- The five-year CSS was 52% for the uterus, 73% for the bladder, and 82% for the extremity/trunk LMS.
- For LMS at all sites, uterine location (HR = 2.14, P < 0.001) and increasing tumour size (HR = 1.05, P < 0.001) were found to be significant predictors of worse CSS on multivariate analysis.
- For urinary bladder LMS, increasing tumour size (HR = 1.18, P = 0.003) was an independent prognostic factor and the conventional staging cut-off threshold of 5 cm for sarcomas outside the head/neck showed statistical significance in stratifying patient risk of cancer-related death.
- Urinary bladder LMS appeared to have clinical behaviour intermediate between those of the extremities/trunk and uterus.
Benjamin et al. [51] concluded that they had suggested that the conventional sarcoma staging protocols based on tumour size should be applied to LMS of the urinary bladder.
Slaoui et al. [52] stated that cases of urinary bladder leiomyosarcoma represent 0.1% of all nonurothelial tumours. Slaoui et al. [52] presented a case report of a 73-year-old man who had undergone a radical cystoprostatectomy for a high-grade urinary bladder leiomyosarcoma with an ileal diversion. The patient recovered uneventfully, and no surgical margins were verified in final pathology. His early follow-up assessments at 3 months demonstrated no signs of computed tomography recurrence and adequate adaptation to his ileal diversion. Slaoui concluded that:
- Even though urinary bladder sarcomas were once thought to have a grim prognosis, recent studies have indicated that adequate surgical treatment can achieve optimal cancer control outcomes.
Nelius et al. [53] in 2010, stated the following: Leiomyosarcomas are a relatively rare tumour entity which is encountered within the urinary bladder. There were just over 100 cases of leiomyosarcomas of the urinary bladder reported in the medical literature by 2010. They had described a case of leiomyosarcoma manifesting initially as a urinary tract infection with lower abdominal pain. A life-threatening episode of visible haematuria had guided the managing clinicians to the final diagnosis and treatment. Due to the rarity of this malignancy, they had presented the case.
Conclusions
- Leiomyosarcoma of the urinary bladder is very rare.
- The majority of clinicians all over the world would not have encountered a patient with primary leiomyosarcoma of the urinary bladder before, in view of the rarity of the neoplasm.
- Interested clinicians and patients would, due to the rarity of primary leiomyosarcoma of the urinary bladder, have to undertake a troublesome search for treatment and outcome information.
- Leiomyosarcomas of the urinary bladder (LMS-UB) are usually aggressive neoplasms.
- Owing to their rarity, only a limited number of cases with clinical follow-up information have been published in the literature.
- There is no current consensus opinion on LMS-UB grading, and it is not known whether the widely accepted Fédération Nationale des Centres de Lutte Contre le Cancer (FNCLCC) and National Cancer Institute (NCI) grading systems of soft-tissue sarcomas apply to LMS-UB.
- Clinicians all over the world who encounter and treat patients who have been diagnosed as having primary leiomyosarcoma of the urinary bladder should publish their experience pursuant to obtaining permission to publish the cases, so that more lessons would be learnt about the neoplasm.
Conflict Of Interest – Nil
Acknowledgements
- World Journal of Surgical Oncology and BMC for granting permission for reproduction of contents and figures from their journal article under the Creative Commons Attribution License under the ensuing copyright statement: Copyright ©2010 Ricciardi et al; licensee BioMed Central Ltd.
- This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
References
- Hamadalla NY, Rifat UN, Safi KC, Mohammed M, Abu-Farsakh H, (2013). Leiomyosarcoma of the urinary bladder: A review and a report of two further cases, Arab Journal of Urology, 11(2), 159–164.
View at Publisher | View at Google Scholar - Ricciardi E, Maniglio P, Schimberni M, Moscarini M, (2010). High-grade leiomyosarcoma of the bladder with delayed onset and very poor prognosis, World Journal of Surgical Oncology, 8, 16.
View at Publisher | View at Google Scholar - Rane S, (2024). Leiomyosarcoma, PathologyOutlines.com.
View at Publisher | View at Google Scholar - Rodríguez D, Barrisford GW, Sanchez A, Preston MA, Kreydin EI, et all., (2014). Primary spermatic cord tumors: Disease characteristics, prognostic factors, and treatment outcomes, Urologic Oncology, 32(1), 52.e19–52.e25.
View at Publisher | View at Google Scholar - Lopes RI, Leite KR, Lopes RN, (2006). Paratesticular leiomyosarcoma treated by enucleation, International Brazilian Journal of Urology, 32(1), 66–67.
View at Publisher | View at Google Scholar - Korkes F, Castro MG, Romero FR, Godoy G, Amary MF, Fernandes RC, Perez MD, (2009). Paratesticular sarcomas in Brazil, Urologia Internationalis, 82(4), 448–452.
View at Publisher | View at Google Scholar - Fisher C, Goldblum JR, Epstein JI, Montgomery E, (2001). Leiomyosarcoma of the paratesticular region: A clinicopathologic study, American Journal of Surgical Pathology, 25(9), 1143–1149.
View at Publisher | View at Google Scholar - Varzaneh FE, Verghese M, Shmookler BM, (2002). Paratesticular leiomyosarcoma in an elderly man, Urology, 60(6), 1112.
View at Publisher | View at Google Scholar - Yuen VT, Kirby SD, Woo YC, (2011). Leiomyosarcoma of the epididymis: Two cases and review of the literature, Canadian Urological Association Journal, 5(6), E121–E124.
View at Publisher | View at Google Scholar - Banowsky LH, Shultz GN, (1970). Sarcoma of the spermatic cord and tunics: Review of the literature and case report, Journal of Urology, 103(5), 628–631.
View at Publisher | View at Google Scholar - Kyratzi I, Lolis E, Antypa E, Lianou MA, Exarhos D, (2011). Imaging features of a huge spermatic cord leiomyosarcoma: Review of the literature, World Journal of Radiology, 3(4), 114–119.
View at Publisher | View at Google Scholar - Blitzer PH, Dosoretz DE, Proppe KH, Shipley WU, (1981). Treatment of malignant tumors of the spermatic cord: A study of 10 cases,
View at Publisher | View at Google Scholar - Fagundes MA, Zietman AL, Althausen AF, Coen JJ, Shipley WU, (1996). The management of spermatic cord sarcoma, Cancer, 77(9), 1873–1876.
View at Publisher | View at Google Scholar - Catton CN, Cummings BJ, Fornasier V, O'Sullivan B, Quirt I, et all., (1991). Adult paratesticular sarcomas: A review of 21 cases, Journal of Urology, 146(2), 342–345.
View at Publisher | View at Google Scholar - Konety BR, Singh J, Lyne JC, Salup RR, (1996). Leiomyosarcoma with osteoclast-like giant cells of the spermatic cord: A case report and review of the literature, Urologia Internationalis, 56(4), 259–262.
View at Publisher | View at Google Scholar - Rubin BP, Fletcher CD, (2000). Myxoid leiomyosarcoma of soft tissue, an underrecognized variant, American Journal of Surgical Pathology, 24(7), 927–936.
View at Publisher | View at Google Scholar - Hornick JL, Fletcher CD, (2003). Criteria for malignancy in non-visceral smooth muscle tumours, Annals of Diagnostic Pathology, 7(1), 60–66.
View at Publisher | View at Google Scholar - Saidani B, Saadi A, Bedoui MA, Zaghbib S, Chakroun M, et all., (2023). Leiomyosarcoma of the bladder: A review and a report of four further cases, International Journal of Surgery Case Reports, 110, 108735.
View at Publisher | View at Google Scholar - Hamadalla NY, Rifat UN, Safi KC, Mohammed M, Abu-Farsakh H, (2013). Leiomyosarcoma of the urinary bladder: A review and a report of two further cases, Arab Journal of Urology, 11(2), 159–164.
View at Publisher | View at Google Scholar - Zieschang H, Koch R, Wirth MP, Froehner M, (2019). Leiomyosarcoma of the urinary bladder in adult patients: A systematic review and meta-analysis, Urologia Internationalis, 102(1), 96–101.
View at Publisher | View at Google Scholar - Tanguay C, Harvey I, Houde M, Srigley JR, Têtu B, (2003). Leiomyosarcoma of urinary bladder following cyclophosphamide therapy: Report of two cases, Modern Pathology, 16(5), 512–514.
View at Publisher | View at Google Scholar - Pedersen-Bjergaard J, Jønsson V, Pedersen M, Hou-Jensen K, (1995). Leiomyosarcoma of the urinary bladder after cyclophosphamide, Journal of Clinical Oncology, 13(2), 532–533.
View at Publisher | View at Google Scholar - Parekh DJ, Jung C, O'Conner J, Dutta S, Smith ER Jr, (2002). Leiomyosarcoma in urinary bladder after cyclophosphamide therapy for retinoblastoma and review of bladder sarcomas, Urology, 60(1), 164.
View at Publisher | View at Google Scholar - Berkmen F, Celebioğlu AS, (1997). Adult genitourinary sarcomas: A report of seventeen cases and review of the literature, Journal of Experimental & Clinical Cancer Research, 16(1), 45–48.
View at Publisher | View at Google Scholar - Dahm P, Gschwend JE, (2003). Malignant non-urothelial neoplasms of the urinary bladder: A review, European Urology, 44(6), 672–681.
View at Publisher | View at Google Scholar - Seo IS, Clark SA, McGovern FD, Clark DL, Johnson EH, (1985). Leiomyosarcoma of the urinary bladder 13 years after cyclophosphamide therapy for Hodgkin's disease, Cancer, 55(7), 1597–1603.
View at Publisher | View at Google Scholar - Rowland RG, Eble JN, (1983). Bladder leiomyosarcoma and pelvic fibroblastic tumor following cyclophosphamide therapy, Journal of Urology, 130(2), 344–346.
View at Publisher | View at Google Scholar - Venkatraman L, Goepel JR, Steele K, Dobbs SP, Lyness RW, et all., (2003). Soft tissue, pelvic, and urinary bladder leiomyosarcoma as second neoplasm following hereditary retinoblastoma, Journal of Clinical Pathology, 56(3), 233–236.
View at Publisher | View at Google Scholar - Kawamura J, Sakurai M, Tsukamoto K, Tochigi H, (1993). Leiomyosarcoma of the bladder eighteen years after cyclophosphamide therapy for retinoblastoma, Urologia Internationalis, 51(1), 49–53.
View at Publisher | View at Google Scholar - Rosser CJ, Slaton JW, Izawa JI, Levy LB, Dinney CP, (2003). Clinical presentation and outcome of high-grade urinary bladder leiomyosarcoma in adults, Urology, 61(6), 1151–1155.
View at Publisher | View at Google Scholar - Mackenzie AR, Whitmore WF Jr, Melamed MR, (1968). Myosarcomas of the bladder and prostate, Cancer, 22(4), 833–844.
View at Publisher | View at Google Scholar - Spiess PE, Kassouf W, Steinberg JR, Tuziak T, Hernandez M, et all., (2007). Review of the M.D. Anderson experience in the treatment of bladder sarcoma, Urologic Oncology, 25(1), 38–45.
View at Publisher | View at Google Scholar - De Berardinis E, Giulianelli R, Zarrelli G, De Santis C, Ginepri A, et all., (1997). Leiomyosarcoma of urinary bladder: Personal experience in 3 cases over a 10-year period, Archivio Italiano di Urologia e Andrologia, 69(Suppl 1), 73–80.
View at Publisher | View at Google Scholar - Strander H, Turesson I, Cavallin-Ståhl E, (2003). A systematic overview of radiation therapy effects in soft tissue sarcomas, Acta Oncologica, 42(5–6), 516–531.
View at Publisher | View at Google Scholar - Swartz DA, Johnson DE, Ayala AG, Watkins DL, (1985). Bladder leiomyosarcoma: A review of 10 cases with 5-year follow-up, Journal of Urology, 133(2), 200–202.
View at Publisher | View at Google Scholar - Martin SA, Sears DL, Sebo TJ, Lohse CM, Cheville JC, (2002). Smooth muscle neoplasms of the urinary bladder: A clinicopathologic comparison of leiomyoma and leiomyosarcoma, American Journal of Surgical Pathology, 26(3), 292–300.
View at Publisher | View at Google Scholar - Cody HS III, Turnbull AD, Fortner JG, Hajdu SI, (1981). The continuing challenge of retroperitoneal sarcomas, Cancer, 47(9), 2147–2152.
View at Publisher | View at Google Scholar - Dotan ZA, Tal R, Golijanin D, Snyder ME, Antonescu C, et all. (2006). Adult genitourinary sarcoma: The 25-year Memorial Sloan-Kettering experience, Journal of Urology, 176(5), 2033–2038.
View at Publisher | View at Google Scholar - O'Sullivan B, Ward I, Catton C, (2003). Recent advances in radiotherapy for soft-tissue sarcoma, Current Oncology Reports, 5(4), 274–281.
View at Publisher | View at Google Scholar - Nelius T, Stevens J, Samathanam C, Filleur S, (2010). Leiomyosarcoma of the urinary bladder presenting as life-threatening gross hematuria, Medical Oncology, 27(2), 562–567.
View at Publisher | View at Google Scholar - Fakhri N, Bhalla R, (2020). Leiomyosarcoma of urinary bladder: A review of two cases, American Journal of Clinical Pathology, 154(Supplement_1), S52.
View at Publisher | View at Google Scholar - Brucker B, Ernst L, Meadows A, Zderic S, (2006). A second leiomyosarcoma in the urinary bladder of a child with a history of retinoblastoma 12 years following partial cystectomy, Pediatric Blood & Cancer, 46(7), 811–814.
View at Publisher | View at Google Scholar - Al-Zahrani AA, Kamal BA, Eldarawani HM, Hashim TM, (2006). Leiomyosarcoma of the bladder in a 16-year-old girl with a history of cyclophosphamide therapy for bilateral retinoblastoma during infancy, Saudi Medical Journal, 27(4), 531–533.
View at Publisher | View at Google Scholar - Motta L, Porcaro AB, Ficarra V, D'Amico A, Piubello Q, et all., (2001). Leiomyosarcoma of the bladder fourteen years after cyclophosphamide therapy for retinoblastoma, Scandinavian Journal of Urology and Nephrology, 35(3), 248–249.
View at Publisher | View at Google Scholar - Kawamura J, Sakurai M, Tsukamoto K, Tochigi H, (1993). Leiomyosarcoma of the bladder eighteen years after cyclophosphamide therapy for retinoblastoma, Urologia Internationalis, 51(1), 49–53.
View at Publisher | View at Google Scholar - Ramírez Sevilla C, Admella-Salvador C, Romero-Martin JA, Llopis-Manzanera J, Barranco-Sanz MA, (2018). Bladder leiomyosarcoma 25 years after treatment with cyclophosphamide in patient with history of retinoblastoma, Urologia Internationalis, 100(1), 119–121.
View at Publisher | View at Google Scholar - Minagawa T, Okaneya T, Kamigaito M, Nishizawa S, Ogawa T, et all., (2008). Leiomyosarcoma of the urinary bladder in a patient with bilateral retinoblastoma, International Journal of Urology, 15(6), 548–550.
View at Publisher | View at Google Scholar - Kin T, Aizawa T, Namiki K, Noda K, Oyama H, Samesima T, et all., (2000). Epithelial membrane antigen-positive leiomyosarcoma of the urinary bladder, Hinyokika Kiyo, 46(3), 189–191.
View at Publisher | View at Google Scholar - Labanaris AP, Zugor V, Meyer B, Nützel R, Helmus S, et all., (2008). Urinary bladder leiomyosarcoma in adults, International Urology and Nephrology, 40(2), 311–316.
View at Publisher | View at Google Scholar - Lindberg MR, Fisher C, Thway K, Cao D, Cheville JC, Folpe AL, (2010). Leiomyosarcoma of the urinary bladder: A clinicopathological study of 34 cases, Journal of Clinical Pathology, 63(8), 708–713.
View at Publisher | View at Google Scholar - Coiner BL, Cates J, Kamanda S, Giannico GA, Gordetsky JB, (2021). Leiomyosarcoma of the urinary bladder: A SEER database study and comparison to leiomyosarcomas of the uterus and extremities/trunk, Annals of Diagnostic Pathology, 53, 151743.
View at Publisher | View at Google Scholar - Slaoui H, Sanchez-Salas R, Validire P, Barret E, Rozet F, et all., (2014). Urinary bladder leiomyosarcoma: Primary surgical treatment, Urology Case Reports, 2(4), 137–138.
View at Publisher | View at Google Scholar - Nelius T, Stevens J, Samathanam C, Filleur S, (2010). Leiomyosarcoma of the urinary bladder presenting as life-threatening gross hematuria, Medical Oncology, 27(2), 562–567.
View at Publisher | View at Google Scholar

 Clinic
Clinic
