Research Article | DOI: https://doi.org/10.31579/2834-8486/012
Diisononyl Phthalate Perturbs Immune Cell Energy Transduction by Disrupting Some Splenic and Lymphocyte Energy Metabolising Enzymes Activities
1Department of Biomedical Science, Faculty of Medicine, and Health Science, Universiti, Putra Malysia.
2Department of Environmental Health Science, Faculty of Basic Medical Sciences, Ajayi Crowther University, Oyo.
*Corresponding Author: Abosede Temitope Olajide., Department of Biomedical Science, Faculty of Medicine, and Health Science, Universiti, Putra Malysia.
Citation: Abosede T Olajide., Samuel A Kehinde., (2024). Diisononyl Phthalate Perturbs Immune Cell Energy Transduction by Disrupting Some Splenic and Lymphocyte Energy Metabolising Enzymes Activities. International Journal of Clinical Therapeutics. 3(3); DOI:10.31579/2834-8486/012
Copyright: © 2024, Abosede Temitope Olajide. Nguyen, this is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 17 January 2023 | Accepted: 03 May 2023 | Published: 10 June 2024
Keywords: energy metabolism-targeted therapy; diisononyl phthalate; spleen; lymphocyte; glycolysis; oxidative phosphorylation
Abstract
Objectives
Energy homeostasis is a hallmark of cell survival and maintenance of cell function, some cell types (e.g., immune cells such as the spleen and lymphocyte) have special functions that demand energy over and above the basic rate (e.g., for antigen processing and presentation, migration and phagocytosis). Independently of the amount of energy needed by cells or the kind of nutrients they metabolize to generate energy, the immediate donor of free energy is mainly represented by adenosine-5’-triphosphate (ATP). The effect of diisononyl phthalate (DiNP) on splenic and lymphocyte energy metabolism was examined.
Methods
Eighteen Wistar rats were divided into 3 groups of six rats each: Group A received Tween-80 (control), DiNP (20 mg/kg/BW) was given to Group B, and 200mg/kg DiNP was given to Group C orally (gavage) for 14 days. The activity of splenic and lymphocyte glycolytic, tricarboxylic acid cycle and oxidative phosphorylation enzymes were assessed.
Results
The glycolytic, tricarboxylic acid cycle and oxidative phosphorylation enzymes studied were predominantly downregulated, except for splenic and lymphocyte lactate dehydrogenase activity at 20mg/kg DiNP relative to control (P < 0.05). DiNP exposure was found to impair splenic and lymphocyte energy transduction enzymes.
Conclusion
Our findings call the need for studying the key energy metabolism pathways and enzymes for diagnosis, disease activity monitoring, guidance for energy metabolism-targeted therapy and monitoring systemic inflammation.
1.Introduction
The most popular plasticizers are phthalates (PAEs), which are added to polyvinyl chloride (PVC) to increase flexibility. Phthalate molecules contaminate the atmosphere, foods, or bodily fluids, exposing both people and the environment they live in (Oje et al., 1997; Liu et al., 2009). Phthalates have been frequently implicated in reproductive issues (Kehinde et al., 2022), cardiac effects (Kehinde et al., 2022), neurological problems, and carcinogenic effects, but reports linking them to some allergic conditions have drawn more attention to their immunotoxicology (Li et al., 2012; Tonk et al., 2012). Diisononyl phthalate (DINP) is a plasticizer that softens polyvinyl chloride (PVC) materials utilise in place of di-2-ethylhexyl phthalate (DEHP). Through eating, inhalation, and skin contact, humans are exposed to DINP. Over 90% of all DINP intake is thought to occur through ingestion, making it the primary route (Chen et al., 2012).
Immunomodulation is an important approach for raising the body's defences against a range of disorders. It means that the immune system is being activated broadly and implies that the creation of effector molecules and the actions of macrophages, natural killer (NK) cells, granulocytes, complement, and lymphocytes are being stimulated without the need for an antigen (Dapas et al., 2014). Spleen, a secondary lymphoid organ is involved in blood storage, the immunological monitoring of bloodborne antigens, and the eradication of old or aberrant blood cells. Additionally, the spleen shelters and aids in the maturation of immune system cells known as lymphocytes. White blood cells called lymphocytes offer defence against pathogens that have managed to infect body cells. By regulating diseased cells, lymphocytes also defend the body against malignant cells. The immune response to infections and antigens in the blood benefits from the spleen (MacIver et al., 2013). T, B, and natural killer (NK) cells are examples of lymphocytes, which are white blood cells that have a uniform appearance but perform a variety of functions (La Motte-Mohs et al., 2005).
In this study, the energy metabolism of the lymphocytes and spleen of rats exposed to DiNP was examined. This is the first investigation into how DiNP affects the splenic and lymphocyte glycolytic, tricarboxylic acid cycle, and oxidative phosphorylation enzymes in DiNP-treated rats.
2.Methods
2.1 Chemicals and assay kits
DiNP was purchased from Relonchem Ltd. (Cheshire, United Kingdom). Lactate dehydrogenase test kit was procured from CYPRESS® Diagnostics (Langdrop, Belgium). Mannitol, sorbitol, sucrose, glucose- 6-phosphate dehydrogenase, ethylenediaminetetraacetic acid (EDTA), nicotinamide adenine dinucleotide (NADH), fructose-1,6-bisphosphate, succinate, phosphoenolpyruvate, oxaloacetate, rotenone, and all other reagents used were of the purest analytical grade and purchased from Sigma- Aldrich (Missouri, USA).
2.2 Experimental animals
At the animal house of the University of Ibadan College of Medicine in Ibadan, 18 male Wistar rats (8 weeks) weighing 200–220g were purchased. The rats were kept in plastic cages at the Ajayi Crowther University animal house for acclimation and treatment. Acclimatization was done for a period of 7 days and rats were allowed unlimited access to pelletized food and water.
2.3 Study design
Three groups of six rats each were created from the rats: DiNP was prepared with ordinary saline and Tween-80 (1:1 v/v). The doses of 20 and 200 mg/kg/day of DiNP were chosen based on prior studies (Kehinde et al., 2022). Group A was given tween/saline as a control, Group B received DiNP (20 mg/kg/BW), and Group C received 200 mg/kg of DiNP. All administrations were carried out over a 14-day period via oral galvage. Tween 80 was utilised as a co-solvent, but it has no impact on the outcomes of the experiments.
2.4 Collection of samples
2.4.1 Isolation of spleen
Animals were euthanized after the last treatment, and the spleen was removed while the animals were under anaesthesia. The removed spleen was cleaned in 1.15% KCL (ice-cold), blotted, weighed, and then homogenised in 10% weight/volume of 0.1 M phosphate-buffered saline (PBS; pH 7.4; 12,000 x g for 10 minutes at 4°C). The supernatant was used for biochemical tests.
2.4.2 Isolation of lymphocytes and mitochondrial fraction
Mitochondria were processed and extracted from the spleen of rats (Erika et al., 2010). Soon after blood collection through cardiac puncture, an aliquot of blood was used to isolate lymphocytes by differential centrifugation using Ficoll paque (Jaatinen and Laine, 2007) and samples were kept at 4°C until the assay was started.
2.4.3 Determination of splenic and lymphocyte glycolytic enzymes activity
The Colowick method was used to determine the activity of hexokinase (Colowick, 1973). According to published method by Sims and Blass (Sims and Blass, 1986), the phosphofructokinase (PFK) activity was determined using the method of Sims and Blass (1986). The method reported by Jagannathan et al. (Jagannathan et al., 1956) was used to measure the activity of aldolase (ALD). As recommended by the manufacturer, the LDH Kit was used to assay the lactate dehydrogenase (LDH) activity (CYPRESS). Tatsumo et al. method was followed to measure NADase activity (Tatsumo et al., 2007).
2.4.4 Determination of splenic and lymphocyte tricarboxylic acid cycle enzymes activity
Citrate synthase activity was assessed using the spectrophotometric enzyme test methodology published by Yu et al. (2007). As previously described by Romkina and Kiriukhin (2017), the isocitrate dehydrogenase (IDH) activity was measured. Using the ????-KGDH Kit and following the manufacturer's (BioVision Incorporated) instructions, the activity of ????-KGDH was assessed. According to Lopez-Calcagno et al. (Lopez-Calcagno et al., 2009), MDH activity was assessed.
2.4.5 Determination of splenic and electron transport chain enzymes activity
According to Medja et al. (2009), Complex I (NADH ubiquinone oxidoreductase), complex II (succinate ubiquinone oxidoreductase, complex III (cytochrome c oxidoreductase) and Complex IV (Cytochrome C Oxidase) activities weas determined.
2.4.6 Total protein determination
The amount of total protein in spleen and lymphocytes was measured using the Gornall et al. (1949).
2.5 Statistical analysis
Results are presented as mean ±SD. The level of homogeneity between the groups was assessed using analysis of variance (ANOVA). When there was heterogeneity, Tukey's test was employed to divide the groups. All analyses were performed using GraphPad Prism® version 8. P values lower than 0.05 were considered statistically significant.
3.Result
3.1 DiNP perturbs splenic and lymphocyte glycolytic enzymes activities
Table 1 displays how DiNP affected the rat spleen and lymphocyte glycolytic enzyme activity. The activities of splenic and lymphocyte HK, ALD, PFK, and NADase were significantly (P <0>
3.2 DiNP downregulates splenic and lymphocyte tricarboxylic acid cycle enzymes activities
The impact of DiNP on the splenic and lymphocyte tricarboxylic acid cycle enzyme activities in the study animals is shown in Figure 2 (A–H). According to the findings, splenic and lymphocyte CS, IDH, -KDH, and MDH activities significantly decreased when compared to control (P < 0>
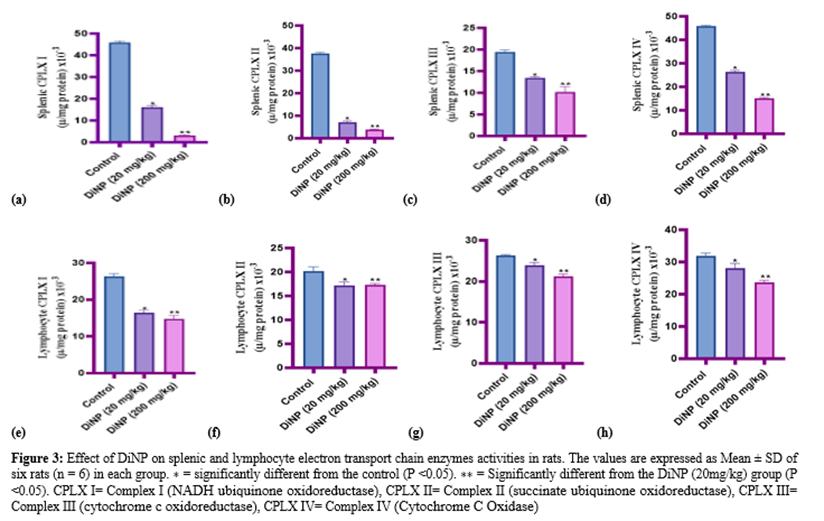
3.3 DiNP downregulates splenic and lymphocyte electron transport chain enzymes activities
Figure 3 (A-H) illustrates how DiNP affected the activity of splenic and lymphocyte electron transport chain enzymes in the study animals. According to the findings, splenic and lymphocyte CPLX I, CPLX II, CPLX III, and CPLX IV activity significantly decreased when compared to control (P < 0> GROUPS SP HK (µ/mg protein) x10-3 SP ALD (µ/mg protein) x10-3 SP PFK (µ/mg protein) x10-3 SP LDH (µ/mg protein) x10-3 SP NADase (µ/mg protein) x10-3 LYM HK (µ/mg protein) x10-3 LYM ALD (µ/mg protein) x10-3 LYM PFK (µ/mg protein) x10-3 LYM LDH (µ/mg protein) x10-3 LYM NADase (µ/mg protein) x10-3 Control 16.13 ± 0.89 28.18 ± 0.59 14.63 ± 0.61 18.72 ± 4.45 2.29 ± 0.21 3.93 ± 0.54 26.48 ± 0.59 23.96 ± 1.79 251.61 ± 12.48 0.47 ± 0.02 DiNP(20mg/kg) 12.52 ±0.77* 19.95 ± 1.34* 13.72 ± 0.89* 30.65 ± 2.26* 1.60 ± 0.27* 0.57 ± 0.09* 18.45 ± 0.62* 20.13 ± 0.58* 266.02 ± 13.06* 0.64 ± 0.03* DiNP(200mg/kg) 7.69± 0.63** 17.28 ± 1.43** 9.40 ± 1.24** 14.38 ± 1.39** 1.26 ± 0.08** 0.22 ± 0.06** 12.61 ± 0.78** 14.22 ± 1.08** 90.78 ± 3.34** 0.55 ± 0.09**
Table 1: Effect of DiNP on Splenic and Lymphocyte Glycolytic enzymes.
* Significantly different when compared with control (P < 0>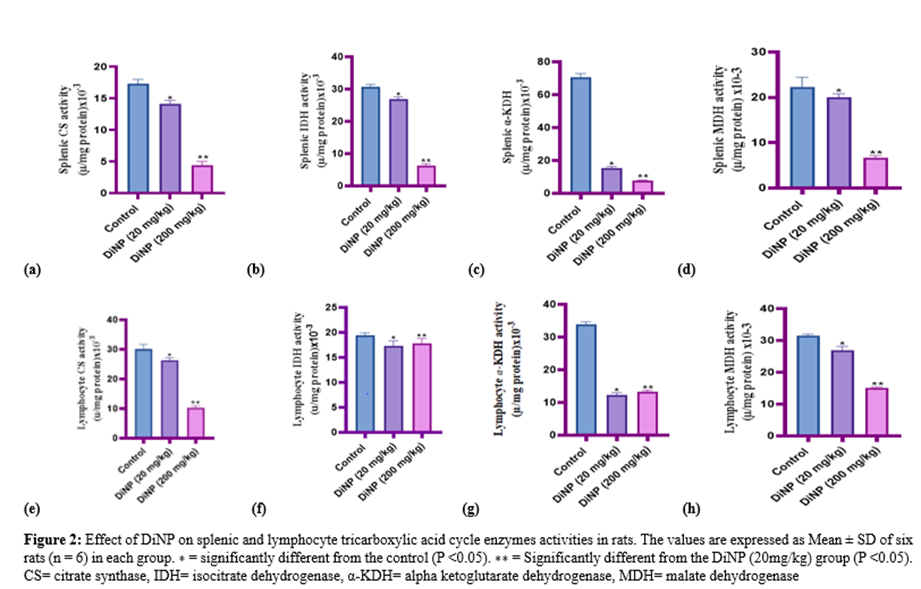
4.Discussion
Cell survival and maintenance of function are characterised by energy homeostasis, yet some cell types (such as immune cells) have specialised tasks that require energy above the basic rate (e.g., for antigen processing and presentation, migration and phagocytosis). Adenosine-5'-triphosphate (ATP) is the primary immediate provider of free energy, regardless of the amount of energy required by cells or the types of substances they metabolise to produce energy (Chaban et al., 2014). This study uses rats as model and offers the first information regarding the spleen and lymphocyte's state of energy metabolism following exposure to DiNP.
Highlights For Review
- Diisononyl phthalate (DiNP) peturbs enzymes of the splenic and lymphocyte glycolytic pathway in Wistar rats.
- Diisononyl phthalate (DiNP) down-regulated enzymes of the splenic and lymphocyte tricarboxylic acid cycle in Wistar rats.
- Energy metabolism pathways may be a useful tool for illness monitoring, diagnosis, energy metabolism-targeted therapy, and systemic inflammatory monitoring.
Because they have an impact on how the immune system reacts to pathogenic bacteria and other foreign substances, lymphocytes are recognised to play a crucial role in the immune system. T cells, B cells, and natural killer cells comprise lymphocytes (Berringto et al., 2005). Different energy metabolism pathways are used by B and T cells depending on the circumstances. The lymph nodes and spleen, which are immune cells, typically function best in compartments with low oxygen (O2) concentrations, or between 0.5 to 4.5% (Caldwell et al., 2001; Donnelly and Finlay, 2015). However, immune cells experience lower O2 concentrations of 0.5–1.0% in pathophysiological circumstances, such as a location of acute and chronic tissue inflammation, which is brought on by an increased oxygen demand resulting from insufficient O2 delivery (Gaber et al., 2005). Lymphocytes are forced to travel against O2 gradients by the hypoxic state mentioned above.
Adenosine triphosphate (ATP), the main immediate supplier of energy for immune processes, comes from aerobic glycolysis and/or oxidative phosphorylation in the mitochondrial respiratory chain, both of which require O2 (Buttgereit etal., 2000). The T cells must rely on lipid oxidation and aerobic glycolysis in hypoxic conditions to meet their energy needs (Semenza, 2012). Activated T cells and B cells have much higher levels of the glycolytic, pentose-phosphate, and glutaminolytic pathways (the glutaminolytic pathway provides a different supply of carbon for the TCA cycle, which converts glutamine to alpha ketoglutarate) (Pearce et al., 2013; Sena et al., 2013). In this investigation, it was discovered that, with the exception of LDH activity, the activities of the glycolytic enzymes (HK, PFK, ALD, and NADase) were downregulated at both DiNP doses (20 mg/kg and 200 mg/kg) in comparison to the control group. This suggests that the glycolysis's final product, pyruvate, won't be available, and as a result, ATP production may be impeded.
B cells (pro and pre) culture, as previously stated require pyruvate from glycolysis to fuel and maintain oxidative phosphorylation (OXPHOS), but the TCA substrates employed are unknown (Yin et al., 2015). However, as compared to the control, the results of this study show that splenic and lymphocyte LDH activities were both elevated at 20mg/kg DiNP. Increased splenic and lymphocyte LDH activity suggests that lactate may be a substrate that feeds the TCA. Lactate is a key substrate for gluconeogenesis and enters the TCA via oxidation of pyruvate and then carboxylation of oxaloacetate. However, it was discovered that splenic and lymphocyte LDH activity was significantly lowered at 200 mg/kg DiNP. This finding shows that as DiNP concentration is raised, LDH activity may be hindered, making the alternative substrate for ATP synthesis unavailable. Furthermore, the enzymes of the succeeding pathways (TCA and OXPHOS) that should have cushioned the energy generation for the spleen and lymphocytes were blocked by the observed decreased splenic and lymphocyte glycolytic enzyme activities as a result of DiNP administration.
T cells and dendritic cells are found in the spleen, where they interact to process antigens and are also directly exposed to cytokines that cause inflammation (Everts and Pearce, 2014). Once activated, T cells' metabolism modifies, and during this increased energy requirement, aerobic glycolysis is utilised (Park and Pan, 2015; Wang et al., 2015; Yin et al., 2016). For systemic inflammatory disease, monitoring spleen energy metabolism has been suggested to be a valuable technique for assessing disease activity and perhaps guiding energy metabolism-targeted therapy (Pak et al., 2014). Inflammation and even impairment of some organs' energy homeostasis have both been linked to DiNP previously using murine models (Adeyemo et al., 2022; Ore et al., 2022). As a result, the spleen and lymphocytes may have undergone inflammation, which would explain why the activity of the glycolytic, TCA, and OXPHOS enzymes have decreased.
Limitation of the Study
The lymphocyte employed for the investigation of energy metabolism was a limitation of this study because it was not differentiated into B or T cells. Using ficoll-paque, an isolated lymphocyte was produced from whole blood.
Conclusion
This research examined how DiNP (20 mg/kg and 200 mg/kg) affected the energy metabolism of lymphocytes and the spleen in rats. The spleen and lymphocytes' energy metabolism were found to be disturbed by DiNP. It was discovered that lactate serves as a substrate for the lymphocyte and spleen TCA cycle. The results of this study may point to important energy metabolism pathways that could be investigated for illness monitoring, diagnosis, energy metabolism-targeted therapy, and systemic inflammatory monitoring.
Author Contributions
Conceptualization: Abosede Temitope Olajide; methodology: Abosede Temitope Olajide and Samuel Abiodun Kehinde; formal analysis, investigation, and resources: Abosede Temitope Olajide and Samuel Abiodun Kehinde; writing- original draft preparation, review and editing: Abosede Temitope Olajide and Samuel Abiodun Kehinde. The final manuscript has been reviewed and approved by the authors.
Funding
Neither this study nor its authors were given a particular grant by funding organisations. The authors paid for their own research.
Declaration of interests
The authors affirm that they have no known financial or interpersonal conflicts that would have appeared to have an impact on the research presented in this study.
References
- Adeyemo et al., 2021. O.A. Adeyemo, A. Ore, E.O. Ajisafe. (2021). The protective effect of piperine on oxidative stress and hepatic damage induced by diisononyl phthalate in rat, Egyptian Journal of Basic and Applied Sciences., 8:1 pp. 293-301.
View at Publisher | View at Google Scholar - Berrington et al., 2005. J.E. Berrington, D. Barge, A.C. Fenton, A.J. Can’t, G.P. Spickett. Lymphocyte subsets in term and significantly preterm UK infants in the first year of life analysed by single platform flow cytometry. Clin Exp Immunol., 140: pp. 289-292.
View at Publisher | View at Google Scholar - F. Buttgereit, G.R. Burmester, M.D. Brand. Buttgerei et al., (2000). Bioenergetics of immune functions: fundamental and therapeutic aspects. Immunol Today., 21(4): pp. 192–199.
View at Publisher | View at Google Scholar - C.C. Caldwell, H. Kojima, D. Lukashev, Caldwell et al., (2001). Differential effects of physiologically relevant hypoxic conditions on T lymphocyte development and effector functions. J Immunol., 167(11): pp. 6140–6149.
View at Publisher | View at Google Scholar - Y. Chaban, E.J. Boekema, N.V. Dudkina. Chaban et al., (2014). Structures of mitochondrial oxidative phosphorylation super complexes and mechanisms for their stabilisation. Biochim Biophys Acta;1837(4): pp. 418–426.
View at Publisher | View at Google Scholar - L. Chen, Y. Zhao, L.X. Li, Chen et al., (2012). Exposure assessment of phthalates in non-occupational populations in China. Sci Total Environ., 427-428, pp. 60-69.
View at Publisher | View at Google Scholar - S.P. Colowick. Colowick, 1973. The Hexokinases, in: The enzymes, 9, Academic Press, pp. 1–48,
View at Publisher | View at Google Scholar - B. Dapas, S. Dall’Acqua, R. Bulla, Dapas et al., (2014). Immunomodulation mediated by an herbal syrup containing a standardized Echinacea root extract: a pilot study in healthy human subjects on cytokine gene expression. Phytomed Int J Phytother Phytopharmacol., 21(11): pp. 1406–1410.
View at Publisher | View at Google Scholar - R.P. Donnelly, D.K. (2015). Finlay. Glucose, glycolysis and lymphocyte responses. Mol. Immunol., 68 pp. 513–519.
View at Publisher | View at Google Scholar - E. Erika Fernández-Vizarra, G. Ferrín, A. Pérez-Martos, P. Fernández-Silva, M. Zeviani, et al. (2010). Isolation of mitochondria for biogenetical studies: an update, Mitochondrion., 10 pp. 253–262.
View at Publisher | View at Google Scholar - Everts and Pearce., B. Everts., E.J. Pearce. (2014). Metabolic control of dendritic cell activation and function: recent advances and clinical implications. Front Immunol., 5: p. 203.
View at Publisher | View at Google Scholar - T. Gaber., R. Dziurla, R. Tripmacher, et al. (2005). Hypoxia inducible factor (HIF) in rheumatology: low O2! See what HIF can do! Ann Rheum Dis.,64(7): pp. 971–980.
View at Publisher | View at Google Scholar - A.G. Gornall, C.J. Bardawill, M.M. David. (1949). Determination of serum proteins by means of the biuret reaction, J. Biol. Chem., 177 pp. 751–766,
View at Publisher | View at Google Scholar - T. Jaatinen, J. Laine. (2007). Isolation of mononuclear cells from human cord blood by Ficoll-Paque density gradient. Curr Protoc Stem Cell Biol., Chapter 2: Unit 2A.1.
View at Publisher | View at Google Scholar - V. Jagannathan, K. Singh, M. Damodaran. (1956). Carbohydrate metabolism in citric acid fermentation. Purification and properties of aldolase from Aspergillus niger, Biochem. J., 63 p. 94.
View at Publisher | View at Google Scholar - S. A. Kehinde., A. Ore., A.T. Olajide., T.P. Fatokun., O.P. Akano. (2022). Diisononyl phthalate negatively perturbs testicular energy metabolism and histoarchitecture of rats. Journal of Hazardous Materials Advances, 100153, ISSN 2772-4166.
View at Publisher | View at Google Scholar - S.A. Kehinde., S.A. A. Ore., A.T. Olajide., I.E. Ajagunna, F.A. Oloyede., T.O. Faniyi., et al. (2022). Diisononyl phthalate inhibits cardiac glycolysis and oxidative phosphorylation by down-regulating cytosolic and mitochondrial energy metabolizing enzymes in murine models. J. Adv. Redox Res., 100041.
View at Publisher | View at Google Scholar - R.N. La Motte-Mohs., E. Herer., J.C. Zuniga-Pflucker. (2005). Induction of T-cell development from human cord blood hematopoietic stem cells by Delta-like 1 in vitro. Blood.,105: pp. 1431-1439.
View at Publisher | View at Google Scholar - X.W. Li, Y. Liang, Y. Su, H. Deng, X.H. Li. (2012). Adverse effects of di-(2-ethylhexyl) phthalate on Leydig cell regeneration in the adult rat testis. Toxicol Lett., 215: pp. 84–91.
View at Publisher | View at Google Scholar - W.L. Liu, C.F. Shen, Z. Zhang, C.B. Zhang. (2009). Distribution of phthalate esters in soil of e-waste recycling sites from Taizhou city in China. Bull Environ Contam Toxico., l82: pp. 665–667.
View at Publisher | View at Google Scholar - P.E. López-Calcagno., J. Moreno., L. Cedeño., L. Labrador., J.L. Concepción., et al. (2009). “Cloning, expression and biochemical characterization of mitochondrial and cytosolic malate dehydrogenase from Phytophthora infestans,”, Mycol. Res., 113 pp. 771–781,
View at Publisher | View at Google Scholar - N.J. MacIver, R.D. Michalek, J.C. (2013). Rathmell. Metabolic regulation of T lymphocytes. Annu Rev Immunol., 31: pp. 259-283.
View at Publisher | View at Google Scholar - F. Medja., S. Allouche., P. Frachon., C. Jardel., M. Malgat., et al., (2009). Development and implementation of standardized respiratory chain spectrophotometric assays for clinical diagnosis, Mitochondrion., 9 pp. 331–339,
View at Publisher | View at Google Scholar - Oie et al., 1997. L. Oie, L.G. Hersoug, J.O. Madsen. R. (1997). esidential exposure to plasticizers and its possible role in the pathogenesis of asthma. Environ Health Perspect., 105: pp. 972–978.
View at Publisher | View at Google Scholar - A. Ore., A.A. Adewale., S.A. Kehinde., T.O. Faniyi., A.D. Oladeji., et al. (2022). Chukwuemeka. Potential roles of oxidative stress and insulin resistance in diisononyl phthalate induced dyslipidemia and hepatosteatosis in BALB/c mice. Advances in Redox Research., 100038.
View at Publisher | View at Google Scholar - K. Pak, S.J. Kim, J.J. Kim, et al. (2014). Splenic FDG uptake predicts poor prognosis in patients with unresectable cholangiocarcinoma. Nuklearmedizin., 53: pp. 26-31.
View at Publisher | View at Google Scholar - B.V. Park, F. Pan. (2015). Metabolic regulation of T cell differentiation and function. Mol Immunol., 68: pp. 497-506.
View at Publisher | View at Google Scholar - E.L. Pearce, M.C. Poffenberger, C.H. Chang et al. (2013). Fueling immunity: insights into metabolism and lymphocyte function. Science., 342: 1242454.
View at Publisher | View at Google Scholar - A.Y. Romkina, M.Y. Kiriukhin, (2017). Biochemical and molecular characterization of the isocitrate dehydrogenase with dual coenzyme specificity from the obligate methylotroph Methylobacillus Flagellatus, Plos one., 12 e0176056.
View at Publisher | View at Google Scholar - G.L. Semenza. (2012). Hypoxia-inducible factors in physiology and medicine. Cell., 148(3): pp. 399–408.
View at Publisher | View at Google Scholar - L.A. Sena, S. Li, A. Jairaman et al. (2013). Mitochondria are required for antigen-specific T cell activation through reactive oxygen species signaling. Immunity., 38: pp. 225–236.
View at Publisher | View at Google Scholar - N.R. Sims, J.B. Blass. (1986). Phosphofructokinase activity in fibroblasts from patients with Alzheimer’s disease and age- and sex matched controls, Metabolic Brain Dis., 1 pp. 83–90.
View at Publisher | View at Google Scholar - I. Tatsuno., J. Sawai., A. Okamoto., M. Matsumoto., M. Minami., et al. (2007) Characterization of the NAD-glycohydrolase in streptococcal strains, Microbiology., 153 pp. 4253–4260,
View at Publisher | View at Google Scholar - E.C. Tonk, A. Verhoef, E.R. E.R. Gremmer, H. van Loveren, A.H. Piersma. (2012). Relative sensitivity of developmental and immune parameters in juvenile versus adult male rats after exposure to di(2-ethylhexyl) phthalate. Toxicol Appl Pharmacol., 260: pp. 48–57.
View at Publisher | View at Google Scholar - H. Wang., T. Li., S. Chen., Y. Gu., S. Ye. (2015). Neutrophil Extracellular Trap Mitochondrial DNA and Its Autoantibody in Systemic Lupus Erythematosus and a Proof-of-Concept Trial of Metformin. Arthritis Rheumatol., 67: pp. 3190-3200.
View at Publisher | View at Google Scholar - Y. Yin, S.C. Choi, Z. Xu, et al. (2015). Normalization of CD4+ T cell metabolism reverses lupus. Sci Transl Med.,7: 274ra218.
View at Publisher | View at Google Scholar - Y. Yin, S.C. Choi, Z. Xu, et al. (2016). Glucose Oxidation Is Critical for CD4+ T Cell Activation in a Mouse Model of Systemic Lupus Erythematosus. J Immunol., 196: pp. 80-90.
View at Publisher | View at Google Scholar - X. Yu, Y.A. Tesiram., R.A. Towner., A. Abbott., E. Patterson., et al. (2007). Early myocardial dysfunction in streptozotocin-induced diabetic mice: a study using in vivo magnetic resonance imaging (MRI), Cardiovasc Diabetol., 6 pp. 1–8,
View at Publisher | View at Google Scholar

 Clinic
Clinic
