Review Article | DOI: https://doi.org/10.31579/2835-7957/033
The Use of Stem Cells in the Treatment of Animal Diseases and Injuries
- Ziad Ahmad Alabdallah *
Department of Anatomy -Histology and Embryology, Al Furat University, Deirez-ZOR, College of Veterinary, Syria.
*Corresponding Author: Ziad Ahmad Alabdallah, Department of Anatomy -Histology and Embryology, Al Furat University, Deirez-ZOR, College of Veterinary, Syria.
Citation: Ziad A. Alabdallah (2023), The Use of Stem Cells in the Treatment of Animal Diseases and Injuries, Clinical Reviews and Case Reports, 2(4); DOI:10.31579/2835-7957/033
Copyright: © 2023, Ziad Ahmad Alabdallah. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: 17 July 2023 | Accepted: 02 August 2023 | Published: 09 August 2023
Keywords: stem cells; regenerative therapy; injuries; diseases
Abstract
Stem cells are considered a scientific renaissance because of their use in regenerative therapy and diseases in the field of veterinary medicine. Therefore, it is necessary to learn about their types and sources, how to take, preserve, and store them, methods of use, and the mechanism of their impact on injuries and diseases that affect animals that may not respond to conventional treatment.
Introduction
Although stem cells (SCs) are undifferentiated cells, they can change into other cell types to perform a variety of tasks. The therapeutic use of stem cells in veterinary medicine makes use of their capacity to differentiate into various cell types. SCs come from four primary sources: embryos, fetuses, umbilical cords, and adult body cells. Once more, stem cells are divided into totipotent, multipotent, pluripotent, and unipotent categories depending on their capacity to differentiate. Multipotent ASCs are employed in treatments because they are easier to get and grow from a variety of sources, have lower immunogenicity, and don't run the danger of developing teratomas. SCs have been used to treat a variety of issues in animals, including heart problems, diabetes mellitus, tendonitis, ligament defects, wounds, cartilage defects, and spinal injuries. However, due to their dynamic complexity, biology, potential for teratoma formation, and histocompatibility, clinical uses of stem cells are constrained. Despite the fact that many degenerative conditions are difficult to treat because they don't respond to current medications, the therapeutic use of stem cells in veterinary medicine hasn't been generally adopted. This manuscript's goal is to discuss the therapeutic use of stem cells in the regeneration of a wide range of difficult illnesses that are not responding to conventional forms of treatment.
Main text:
Types of stem cells
The two primary types of stem cells used in contemporary regenerative treatment are allogeneic and autologous. Experts throughout the world are constantly debating the differences in the safety and efficacy of using these cells in treatment. Depending on the donor-recipient connection, MSCs can be classified as being of allogeneic, autologous, or xenogeneic origin. The individual receiving allogeneic cells belongs to the same species as the donor. Successful in vitro production of these cells reduces the duration of the therapy significantly as compared to autologous cells. Horses were given therapeutic injections of the antlerogenic stem cells MIC-1 (obtained from a deer), and injured rabbits had the cells experimentally implanted into their tissues. [75].
Isolation and purification of stem cells
Practically any tissue in a living body can be efficiently harvested to produce mesenchymal stem cells. Nevertheless, the best results are obtained when cells are isolated from adult tissues like adipose tissue (AD-MSCs), bone marrow (BM-MSCs), peripheral blood (PB-MSCs), or fetal tissue like the placenta (P-MSCs), umbilical cord blood (UCB-MSCs), or umbilical cord (UC-MSCs) [7;26]. This is a result of these cells' capacity for proliferation and differentiation. It was found that the type of cells, based on their origin, has a significant impact on both their capacity for in vivo differentiation and their physiologically significant properties [100]. When selecting the source of those cells, a crucial factor to take into account is how the MSCs will be used in the therapy. Similar to human medicine, one of the most extensively studied sources of MSC origin in veterinary medicine is bone marrow [39]. Unfortunately, gathering the samples requires an invasive procedure done while the animals are sedated, with or without local anesthetic in the case of dogs, and under general anesthesia in the case of horses. Both of these procedures have a risk of postoperative complications, including infection and/or hemorrhage [73]. A number of significant inadvertent cardio-thoracic punctures have been reported [45]. as well as nonfatal pneumopericardium in horses [29;44]. But with these animals, there is little to no risk of the aforementioned issues when doing the BM-MSC collection procedure on a standing animal. The 4th or 5th sternebra is the best place to perform a sternal biopsy with a Jamshidi needle, according to current consensus [24; 47]. Age-related declines in the number of BM-MSCs, which make up a relatively small percentage of all bone marrow stromal mononuclear cells [77]. The donor's previously removed aspirate is used for MSC passage and culture in vitro. The maximum number of cultivated MSCs that can be passed through four times determines the exact therapeutic amount of MSCs. This culture goes through several stages, including starting the culture, cell growth, media changes, and finishing the culture by identifying the cell phenotype. Despite the fact that research first concentrated on MSCs produced from bone marrow, the large concentration of MSCs in adipose tissues (100–1000 times that in bone marrow) led to the use of adipose-derived MSCs in regenerative stem cell treatment [15;66;109]. The need for numerous purification processes to exclude non-MSC cell types (such as hematopoietic stem cells and blood cells) when collecting chicken MSCs using bone marrow from non-compact bone sources is one of the main problems [48]. These methods of purification frequently result in cytotoxicity, modifications to MSC functioning, and poor MSC yields [103]. There haven't been any reports of MSCs isolated from chicken compact bones, which might circumvent these restrictions because there aren't as many contaminating cell types [74]. Hematopoietic stem cells and MSCs are the two principal stem cell groups that exist in bone marrow [74; 108]. The contamination of blood cells and hematopoietic stem cells that results from the isolation of MSCs from bone marrow is a severe disadvantage. The MSC population extracted from bone marrow has been purified or enriched using a variety of methods, including density gradient centrifugation and preferential attachment to culture plastic [105], antibody-based cell sorting and the application of ficole to remove blood cells [30;97], techniques for low- and high-density cultivation [31], as well as regular media change [88]. However, there are a number of drawbacks to these techniques for purifying MSCs that have been isolated from bone marrow. A phenotypically and functionally heterogeneous cell population might be produced via preferential attachment to cell culture plates [95]. Using immune depletion strategies decreased the expression of several genes involved in cell growth and cell cycle progression [3]. Insulin-like growth factor or leukemia inhibitory factor exposure of MSCs obtained from immunodepleted cells reversibly decreased the cells' capacity to develop into adipocytes, chondrocytes, and osteoblasts in vitro [3]. Only around 27 fibrobalstoid colonies of 5 or more cells were produced using low-density culture out of a total of 200 culture discs [102]. Multiple multi-lineage MSCs were recovered from hematopoietic cells using cell sorting techniques, but these MSCs had less osteogenic capacity and less clonogenicity than unilinear MSCs [97]. MSCs can be easily and cheaply extracted from compact bones, eliminating the need for further purification procedures and lowering the risk of hematopoietic cell contamination in isolated cultures [41;108].
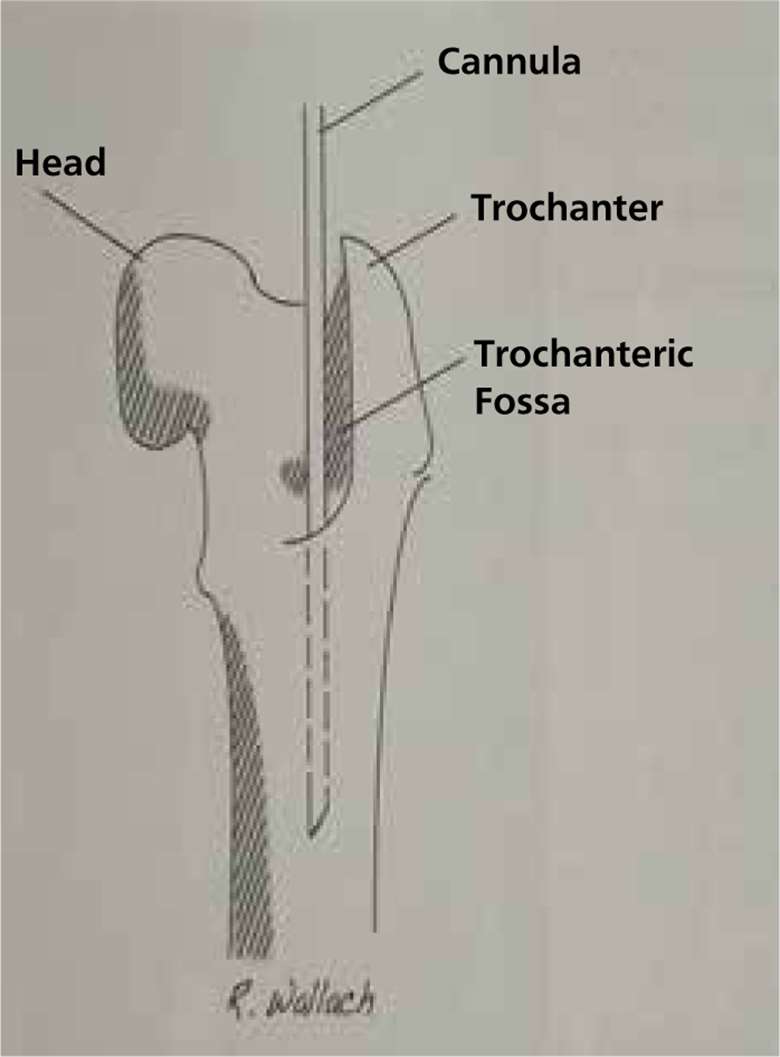 | |
Diagram to show a catheter placed into the proximal femur | Taking stem cells from the pelvic bone in dogs |
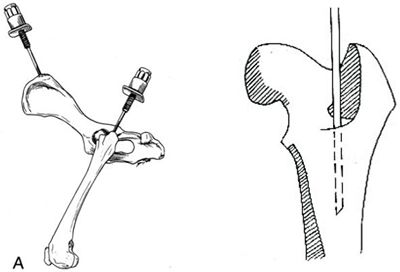 |  |
Taking stem cells from the pelvic bone and femur in dogs | Taking stem cells from the humerus of dogs |
Preservation and storage of stem cells
To get enough MSCs for clinical usage, they must be separated and grown in vitro for therapeutic use. It may occasionally be necessary to apply a second or third time; however, prolonged culture before therapeutic use is not advised since the cells might lose their stemness characteristics and get contaminated with germs. For these reasons, it is highly helpful to cryopreserve these cells in order to obtain a fast and regulated supply of many autologous stem cells that maintain the vitality and pluripotent phenotype of the freshly separated cells while maintaining the unmodified properties of the cells. Di Bella et al. [25] assessed the outcomes of a 7-year cryopreservation procedure utilizing 10% DMSO and various FBS concentrations (ranging from 10% to 90%). In both fresh and thawed cells, the phenotypes of morphology, cell viability, differentiation, and proliferative capacity, as well as the expression of pluripotency markers, were examined. According to the results of this investigation, canine adipose tissue MSCs that were cryopreserved with more than 50
Conclusion:
This review aims to highlight the importance of using stem cells in the treatment of many injuries and diseases in different animals when conventional treatment does not have any effect on the pathological condition and stem cell therapy can be combined with conventional treatment in order for the therapeutic results to be faster, thus preserving the health of the animal and its productive capacity.
Acknowledgment
None.
Acknowledgment
None.
Conflict of Interest
None.
References
- Angelone M., Conti V., Biacca C., Battaglia B., Pecorari L et al. (2017) The contribution of adipose tissue-derived mesenchymal stem cells and platelet-rich plasma to the treatment of chronic equine laminitis: A proof of concept. Int. J. Mol. Sci.18:2122.
View at Publisher | View at Google Scholar - Asari S., Itakura S., Ferreri K., Liu C., Kuroda Y. (2009). Mesenchymal stem cells suppress B-cell terminal differentiation. Exp. Hematol.37:604–615.
View at Publisher | View at Google Scholar - Baddoo, M., Hill, K., Wilkinson, R., Gaupp, D., Hughes, C.et al. (2003). Characterization of mesenchymal stem cells isolated from murine bone marrow by negative selection. J. Cell Biochem. 89, 1235–1249.
View at Publisher | View at Google Scholar - Bahsoun S., Coopman K., Akam E.C. (2019). The impact of cryopreservation on bone marrow-derived mesenchymal stem cells: A systematic review. J. Transl. Med. 17:397.
View at Publisher | View at Google Scholar - Barberini D.J., Aleman M., Aristizabal F., Spriet M., Clark K.C. (2018). Safety and tracking of intrathecal allogeneic mesenchymal stem cell transplantation in healthy and diseased horses. Stem Cell Res. Ther. 9:96.
View at Publisher | View at Google Scholar - Barussi F.C.M., Bastos F.Z., Leite L.M.B., Fragoso F.Y.I., Senegaglia A.C. et al. (2016). Intratracheal therapy with autologous bone marrow-derived mononuclear cells reduces airway inflammation in horses with recurrent airway obstruction. Respir. Physiol. Neurobiol. 2016; 232:35–42.
View at Publisher | View at Google Scholar - Bearden, R.N.; Huggins, S.S.; Cummings, K.J.; Smith, R.; Gregory, C.A.et al. (2017). In-vitro characterization of canine multipotent stromal cells isolated from synovium, bone marrow, and adipose tissue: A donor-matched comparative study. Stem Cell Res. Ther.
View at Publisher | View at Google Scholar - Broeckx, S.; Zimmerman, M.; Crocetti, S.; Suls, M.; Mariën, T et al. (2014). Regenerative therapies for equine degenerative joint disease: A preliminary study.
View at Publisher | View at Google Scholar - Broeckx, S.Y.; Seys, B.; Suls, M.; Vandenberghe, A.; Mariën, T.; Adriaensen, E.; Declercq, J.; Van Hecke, L.; Braun, G.; Hellmann, K.; et al. Equine Allogeneic Chondrogenic Induced Mesenchymal Stem Cells Are an Effective Treatment for Degenerative Joint Disease in Horses. Stem Cells Dev. 2019, 28, 410–422. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Bronzini, I.; Patruno, M.; Iacopetti, I.; Martinello, T. Influence of Temperature, Time and Different Media on Mesenchymal Stromal Cells Shipped for Clinical Application. Vet. J. Lond. Engl. 1997 2012, 194, 121–123. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Caplan, A.I. Mesenchymal Stem Cells: Time to Change the Name! Stem Cells Transl. Med. 2017, 6, 1445–1451. [Google Scholar] [CrossRef] [PubMed][Green Version]
View at Publisher | View at Google Scholar - Carvalho K.A.T., Cury C.C., Oliveira L., Cattaned R.I.I., Malvezzi M., Francisco J.C., Pachalok A., Olandoski M., Faria-Neto J.R., Guarita-Souza L.C. Evaluation of bone marrow mesenchymal stem cell standard cryopreservation procedure efficiency. Transpl. Proc. 2008;40:839–841. doi: 10.1016/j.transproceed.2008.03.004. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Casañas J., de la Torre J., Soler F., Garcia F., Rodellar C., Pumarola M., Climent J., Soler R., Orozco L. Peripheral nerve regeneration after experimental section in ovine radial and tibial nerves using synthetic nerve grafts, including expanded bone marrow mesenchymal cells: Morphological and neurophysiological results. Injury. 2014;45:S2–S6. doi: 10.1016/S0020-1383(14)70003-8. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Chang, L.-B.; Peng, S.-Y.; Chou, C.-J.; Chen, Y.-J.; Shiu, J.-S.; Tu, P.-A.; Gao, S.-X.; Chen, Y.-C.; Lin, T.-K.; Wu, S.-C. Therapeutic Potential of Amniotic Fluid Stem Cells to Treat Bilateral Ovarian Dystrophy in Dairy Cows in a Subtropical Region. Reprod. Domest. Anim. 2018, 53, 433–441. [Google Scholar] [CrossRef] [PubMed][Green Version]
View at Publisher | View at Google Scholar - Choudhery, M.S.; Badowski, M.; Muise, A.; Harris, D.T. Comparison of human mesenchymal stem cells derived from adipose and cord tissue. Cytotherapy 2013, 15, 330–343. [Google Scholar] [CrossRef] [PubMed]
View at Publisher | View at Google Scholar - Cizkova, D.; Cubinkova, V.; Smolek, T.; Murgoci, A.-N.; Danko, J.; Vdoviakova, K.; Humenik, F.; Cizek, M.; Quanico, J.; Fournier, I.; et al. Localized Intrathecal Delivery of Mesenchymal Stromal Cells Conditioned Medium Improves Functional Recovery in a Rat Model of Spinal Cord Injury. Int. J. Mol. Sci. 2018, 19, 870. [Google Scholar] [CrossRef][Green Version]
View at Publisher | View at Google Scholar - Colleoni, S.; Bottani, E.; Tessaro, I.; Mari, G.; Merlo, B.; Romagnoli, N.; Spadari, A.; Galli, C.; Lazzari, G. Isolation, Growth and Differentiation of Equine Mesenchymal Stem Cells: Effect of Donor, Source, Amount of Tissue and Supplementation with Basic Fibroblast Growth Factor. Vet. Res. Commun. 2009, 33, 811. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Corradetti B., Correani A., Romaldini A., Marini M.G., Bizzaro D., Perrini C., Cremonesi F., Lange-Consiglio A. Amniotic membrane-derived mesenchymal cells and their conditioned media: Potential candidates for uterine regenerative therapy in the horse. PLoS ONE. 2014;9:e111324. doi: 10.1371/journal.pone.0111324. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - da Silva Meirelles L., Fontes A.M., Covas D.T., Caplan A.I. Mechanisms involved in the therapeutic properties of mesenchymal stem cells. Cytokine Growth Factor Rev. 2009;20:419–427. doi: 10.1016/j.cytogfr.2009.10.002. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Das, J.; Behera, S.S. Successful treatment of inter digital chronic ulcerative wound by mesenchymal stem cell therapy in a cow. Indian J. Vet. Sci. Biotechnol. 2013, 9, 66–67. [Google Scholar]
View at Publisher | View at Google Scholar - Das, J.; Nath, I.; Das, R.K.; Routray, P.; Behera, S.S. Autologous Stem Cell Therapy to Treat Chronic Ulcer in Heifer-A Case Study. Available online: https://scholar.google.com/citations?view_op=view_citation&hl=en&user=V8kdet8AAAAJ&citation_for_view=V8kdet8AAAAJ:UeHWp8X0CEIC (accessed on 15 July 2021).
View at Publisher | View at Google Scholar - Das, J.; Routray, N.P.; Das, R.K. Autologous Bone Marrow Derived Mesenchymal Stem Cells (BM-MSCs) Therapy for Treatment of Chronic Limb Wound in a Bull. Indian J. Available online: https://www.indianjournals.com/ijor.aspx?target=ijor:ipo&volume=14&issue=1&article=023 (accessed on 9 December 2021).
View at Publisher | View at Google Scholar - de Papa P.M., Guasti P.N., De Vita B., Nakazato N.G., Maia L., de Freitas Dell’Aqua C.P., da Scheeren V.F.C., Segabinazzi L.G.T.M., de Andrade Junior L.R.P., Silva L.F.M.C., et al. Clinical safety of intratesticular transplantation of allogeneic bone marrow multipotent stromal cells in stallions. Reprod. Domest. Anim. 2020;55:429–437. doi: 10.1111/rda.13624. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Désévaux, C.; Laverty, S.; Doizé, B. Sternal bone biopsy in standing horses. Vet. Surg. 2000, 29, 303–308. [Google Scholar]
View at Publisher | View at Google Scholar - Di Bella, S.; Cannella, V.; Mira, F.; Di Marco, P.; Lastra, A.; Gucciardi, F.; Purpari, G.; Guercio, A. The Effect of a 7 Year-Long Cryopreservation on Stemness Features of Canine Adipose-Derived Mesenchymal Stem Cells (CAD-MSC). Animals 2021, 11, 1755. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Ding, D.-C.; Shyu, W.-C.; Lin, S.-Z. Mesenchymal Stem Cells. Cell Transplant. 2011, 20, 5–14. [Google Scholar] [CrossRef] [PubMed][Green Version]
View at Publisher | View at Google Scholar - Dryden V.C., Morrison S., Bras R., Morrell S.A. Using stem cells in clinical cases. J. Equine Vet. Sci. 2013;33:872–873. doi: 10.1016/j.jevs.2013.08.057. [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Duan W., Lopez M.J., Hicok K. Adult multipotent stromal cell cryopreservation: Pluses and pitfalls. Vet. Surg. 2018;47:19–29. doi: 10.1111/vsu.12730. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Durando, M.M.; Zarucco, L.; Schaer, T.P.; Ross, M.; Reef, V.B. Pneumopericardium in a horse secondary to sternal bone marrow aspiration. Equine Vet. Educ. 2006, 18, 75–79. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - El-Sayed, K. M., Paris, S., Graetz, C., Kassem, N., Mekhemar, M., Ungefroren, H., et al. (2015). Isolation and characterisation of human gingival margin-derived STRO-1/MACS(+) and MACS(-) cell populations. Int. J. Oral. Sci. 7, 80–88. doi: 10.1038/ijos.2014.41. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Eslaminejad, M. B., and Nadri, S. (2009). Murine mesenchymal stem cell isolated and expanded in low and high density culture system: surface antigen expression and osteogenic culture mineralization. In vitro Cell Dev. Biol. Anim. 45, 451–459. doi: 10.1007/s11626-009-9198-1.[PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Espina, M.; Jülke, H.; Brehm, W.; Ribitsch, I.; Winter, K.; Delling, U. Evaluation of Transport Conditions for Autologous Bone Marrow-Derived Mesenchymal Stromal Cells for Therapeutic Application in Horses. PeerJ 2016, 4, e1773. [Google Scholar] [CrossRef] [PubMed][Green Version]
View at Publisher | View at Google Scholar - Ferris R.A., Frisbie D.D., McCue P.M. Use of mesenchymal stem cells or autologous conditioned serum to modulate the inflammatory response to spermatozoa in mares. Theriogenology. 2014;82:36–42. doi: 10.1016/j.theriogenology.2014.02.015. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Ferris, D.J.; Frisbie, D.D.; Kisiday, J.D.; Mcllwraith, C.W.; Hague, B.A.; Major, M.D.; Schneider, R.K.; Zubrod, C.J.; Kawcak, C.E.; Goodrich, L.R. Clinical outcome after intra-articular administration of bone marrow derived mesenchymal stem cells in 33 horses with stifle injury. Vet. Surg. 2014, 43, 255–265. [Google Scholar] [CrossRef][Green Version]
View at Publisher | View at Google Scholar - García-Mendívil, L.; Mediano, D.R.; Hernaiz, A.; Sanz-Rubio, D.; Vázquez, F.J.; Marín, B.; López-Pérez, Ó.; Otero, A.; Badiola, J.J.; Zaragoza, P.; et al. Effect of Scrapie Prion Infection in Ovine Bone Marrow-Derived Mesenchymal Stem Cells and Ovine Mesenchymal Stem Cell-Derived Neurons. Animals 2021, 11, 1137. [Google Scholar] [CrossRef] [PubMed]
View at Publisher | View at Google Scholar - Garvican, E.R.; Cree, S.; Bull, L.; Smith, R.K.; Dudhia, J. Viability of Equine Mesenchymal Stem Cells during Transport and Implantation. Stem Cell Res. Ther. 2014, 5, 1. [Google Scholar] [CrossRef][Green Version]
View at Publisher | View at Google Scholar - Ghai, S.; Saini, S.; Ansari, S.; Verma, V.; Chopra, S.; Sharma, V.; Devi, P.; Malakar, D. Allogenic Umbilical Cord Blood-Mesenchymal Stem Cells Are More Effective than Antibiotics in Alleviating Subclinical Mastitis in Dairy Cows. Theriogenology 2022, 187, 141–151. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Ghai, S.; Verma, V.N.; Ansari, S.; Saini, S.; Thakur, A.; Kumar, A.; Kumar, S.; Malakar, D.; Ghai, S.; Verma, V.N.; et al. 157 Mesenchymal Stem Cells as a Regenerative Therapy for the Prevention of Subclinical Mastitis in Cattle. Reprod. Fertil. Dev. 2021, 34, 316–317. [Google Scholar] [CrossRef] [PubMed]
View at Publisher | View at Google Scholar - Godwin, E.E.; Young, N.J.; Dudhia, J.; Beamish, I.C.; Smith, R.K.W. Implantation of bone marrow-derived mesenchymal stem cells demonstrates improved outcome in horses with overstrain injury of the superficial digital flexor tendon. Equine Vet. J. 2012, 44, 25–32. [Google Scholar] [CrossRef] [PubMed]
View at Publisher | View at Google Scholar - Grady S.T., Watts A.E., Thompson J.A., Penedo M.C.T., Konganti K., Hinrichs K. Effect of intra-ovarian injection of mesenchymal stem cells in aged mares. J. Assist. Reprod. Genet. 2019;36:543–556. doi: 10.1007/s10815-018-1371-6. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Guo, Z., Li, H., Li, X., Yu, X., Wang, H., Tang, P., et al. (2006). In vitro characteristics and in vivo immunosuppressive activity of compact bone-derived murine mesenchymal progenitor cells. Stem Cells 24, 992–1000. doi: 10.1634/stemcells.2005-0224.[PubMed][CrossRef][Google Scholar]
View at Publisher | View at Google Scholar - Iacono E., Lanci A., Merlo B., Ricci F., Pirrone A., Antonelli C., Mariella J., Castagnetti C. Effects of amniotic fluid mesenchymal stem cells in carboxymethyl cellulose gel on healing of spontaneous pressure sores: Clinical outcome in seven hospitalized neonatal foals. Turkish J. Biol. 2016;40:484–492. doi: 10.3906/biy-1507-147. [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Iacono E., Merlo B., Pirrone A., Antonelli C., Brunori L., Romagnoli N., Castagnetti C. Effects of mesenchymal stem cells isolated from amniotic fluid and platelet-rich plasma gel on severe decubitus ulcers in a septic neonatal foal. Res. Vet. Sci. 2012;93:1439–1440. doi: 10.1016/j.rvsc.2012.04.008. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Iacono, E.; Lanci, A.; Gugole, P.; Merlo, B. Shipping Temperature, Time and Media Effects on Equine Wharton’s Jelly and Adipose Tissue Derived Mesenchymal Stromal Cells Characteristics. Animals 2022, 12, 1967. [Google Scholar] [CrossRef] [PubMed]
View at Publisher | View at Google Scholar - Jacobs, R.M.; Kociba, G.J.; Ruoff, W.W. Monoclonal Gammopathy in a Horse with Defective Hemostasis. Vet. Pathol. 1983, 20, 643–647. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Karnieli, O.; Friedner, O.M.; Allickson, J.G.; Zhang, N.; Jung, S.; Fiorentini, D.; Abraham, E.; Eaker, S.S.; Yong, T.K.; Chan, A.; et al. A consensus introduction to serum replacements and serum-free media for cellular therapies. Cytotherapy 2017, 19, 155–169. [Google Scholar] [CrossRef] [PubMed]
View at Publisher | View at Google Scholar - Kasashima, Y.; Ueno, T.; Tomita, A.; Goodship, A.E.; Smith, R.K.W. Optimisation of bone marrow aspiration from the equine sternum for the safe recovery of mesenchymal stem cells. Equine Vet. J. 2011, 43, 288–294. [Google Scholar] [CrossRef] [PubMed]
View at Publisher | View at Google Scholar - Khatri, M., O'Brien, T. D., and Sharma, J. M. (2009). Isolation and differentiation of chicken mesenchymal stem cells from bone marrow. Stem Cells Dev. 18, 1485–1492. doi: 10.1089/scd.2008.0223. [PubMed ] [CrossRef ] [Google Scholar]
View at Publisher | View at Google Scholar - Klinger, C. Mesenchymal stem cells: A potential therapy for canine atopic dermatitis? Vet. Rec. 2018, 183, 651. [Google Scholar] [CrossRef] [PubMed]
View at Publisher | View at Google Scholar - La Mantia, D.; Bernardini, C.; Zannoni, A.; Salaroli, R.; Wang, C.; Bencivenni, S.; Forni, M. Efficacy of Stem Cell Therapy in Large Animal Models of Ischemic Cardiomyopathies: A Systematic Review and Meta-Analysis. Animals 2022, 12, 749. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Lalu M.M., Sullivan K.J., Mei S.H.J., Moher D., Straus A., Fergusson D.A., Stewart D.J., Jazi M., Macleod M., Winston B., et al. Evaluating mesenchymal stem cell therapy for sepsis with preclinical meta-analyses prior to initiating a first-in-human trial. Elife. 2016;5:1–28. doi: 10.7554/eLife.17850. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Lanci A., Merlo B., Mariella J., Castagnetti C., Iacono E. Heterologous Wharton’s Jelly derived mesenchymal stem cells application on a large chronic skin wound in a 6-month-old filly. Front. Vet. Sci. 2019;6:9. doi: 10.3389/fvets.2019.00009. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Lima, V.P.; Tobin, G.C.; Jesus Pereira MR de Silveira, M.D.; Witz, M.I.; Nardi, N.B. Chondrogenic effect of liquid and gelled platelet lysate on canine adipose-derived mesenchymal stromal cells. Res. Vet. Sci. 2019, 124, 393–398. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Lin H.-B., Li W.-L., Zhuo X., He A.-Q. Application effect of soft corneal contact lens on corneal limbal stem cell transplantation in elderly patients with pterygium. Int. J. Ophthalmol. 2019;19:329–331. [Google Scholar]
View at Publisher | View at Google Scholar - Maia L., da Cruz Landim-Alvarenga F., Taffarel M.O., de Moraes C.N., Machado G.F., Melo G.D., Amorim R.M. Feasibility and safety of intrathecal transplantation of autologous bone marrow mesenchymal stem cells in horses. BMC Vet. Res. 2015;11:63. doi: 10.1186/s12917-015-0361-5. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Maki, C.B.; Beck, A.; Wallis, C.-B.C.C.; Choo, J.; Ramos, T.; Tong, R.; Borjesson, D.L.; Izadyar, F. Intra-articular Administration of Allogeneic Adipose Derived MSCs Reduces Pain and Lameness in Dogs With Hip Osteoarthritis: A Double Blinded, Randomized, Placebo Controlled Pilot Study. Front. Vet. Sci. 2020, 7, 570. [Google Scholar] [CrossRef] [PubMed]
View at Publisher | View at Google Scholar - Malard, P.F.; Peixer, M.A.S.; Grazia, J.G.; Brunel, H.D.S.S.; Feres, L.F.; Villarroel, C.L.; Siqueira, L.G.B.; Dode, M.A.N.; Pogue, R.; Viana, J.H.M.; et al. Intraovarian Injection of Mesenchymal Stem Cells Improves Oocyte Yield and in Vitro Embryo Production in a Bovine Model of Fertility Loss. Sci. Rep. 2020, 10, 8018. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Mambelli L.I., Mattos R.C., Winter G.H.Z., Madeiro D.S., Morais B.P., Malschitzky E., Miglino M.A., Kerkis A., Kerkis I. Changes in expression pattern of selected endometrial proteins following mesenchymal stem cells infusion in mares with endometrosis. PLoS ONE. 2014;9:e97889. doi: 10.1371/journal.pone.0097889. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Mambelli L.I., Winter G.H.Z., Kerkis A., Malschitzky E., Mattos R.C., Kerkis I. A novel strategy of mesenchymal stem cells delivery in the uterus of mares with endometrosis. Theriogenology. 2013;79:744–750. doi: 10.1016/j.theriogenology.2012.11.030. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Martinello T., Bronzini I., Maccatrozzo L., Mollo A., Sampaolesi M., Mascarello F., Decaminada M., Patruno M. Canine adipose-derived-mesenchymal stem cells do not lose stem features after a long-term cryopreservation. Res. Vet. Sci. 2011;91:18–24. doi: 10.1016/j.rvsc.2010.07.024. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Martinet L., Fleury-Cappellesso S., Gadelorge M., Dietrich G., Bourin P., Fournie J.J., Poupot R. A regulatory cross-talk between Vγ9Vδ2 T lymphocytes and mesenchymal stem cells. Eur. J. Immunol. 2009;39:752–762. doi: 10.1002/eji.200838812. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Marycz K., Szłapka-Kosarzewska J., Geburek F., Kornicka-Garbowska K. Systemic sdministration of rejuvenated adipose-derived mesenchymal stem cells improves liver metabolism in equine metabolic syndrome (EMS)—New approach in veterinary regenerative medicine. Stem Cell Rev. Rep. 2019;15:842–850. doi: 10.1007/s12015-019-09913-3. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Mercati, F.; Pascucci, L.; Curina, G.; Scocco, P.; Tardella, F.M.; Dall’aglio, C.; Marini, C.; Ceccarelli, P. Evaluation of Storage Conditions on Equine Adipose Tissue-Derived Multipotent Mesenchymal Stromal Cells. Vet. J. Lond. Engl. 1997 2014, 200, 339–342. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Morrison S. Successful use of allogenic umbilical cord-derived stem cells in nonresponsive chronic laminitic cases. J. Equine Vet. Sci. 2011;31:603. doi: 10.1016/j.jevs.2011.09.053. [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Mund S.J.K., Kawamura E., Awang-Junaidi A.H., Campbell J., Wobeser B., MacPhee D.J., Honaramooz A., Barber S. Homing and engraftment of intravenously administered equine cord blood-derived multipotent mesenchymal stromal cells to surgically created cutaneous wound in horses: A pilot project. Cells. 2020;9:1162. doi: 10.3390/cells9051162. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Nakao, N.; Nakayama, T.; Yahata, T.; Muguruma, Y.; Saito, S.; Miyata, Y.; Yamamoto, K.; Naoe, T. Adipose tissue-derived mesenchymal stem cells facilitate hematopoiesis in vitro and in vivo: Advantages over bone marrow-derived mesenchymal stem cells. Am. J. Pathol. 2010, 177, 547–554. [Google Scholar] [CrossRef] [PubMed]
View at Publisher | View at Google Scholar - Navarrete F., Saravia F., Cisterna G., Rojas F., Silva P.P., Rodríguez-Alvarez L., Rojas D., Cabezas J., Mançanares A.C.F., Castro F.O. Assessment of the anti-inflammatory and engraftment potential of horse endometrial and adipose mesenchymal stem cells in an in vivo model of post breeding induced endometritis. Theriogenology. 2020;155:33–42. doi: 10.1016/j.theriogenology.2020.06.010. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Nemeth K., Keane-Myers A., Brown J.M., Metcalfe D.D., Gorham J.D., Bundoc V.G., Hodges M.G., Jelinek I., Madala S., Karpati S., et al. Bone marrow stromal cells use TGF-β to suppress allergic responses in a mouse model of ragweed-induced asthma. Proc. Natl. Acad. Sci. USA. 2010;107:5652–5657. doi: 10.1073/pnas.0910720107. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Pacini, S.; Spinabella, S.; Trombi, L.; Fazzi, R.; Galimberti, S.; Dini, F.; Carlucci, F.; Petrini, M. Suspension of bone marrow-derived undifferentiated mesenchymal stromal cells for repair of superficial digital flexor tendon in race horses. Tissue Eng. 2007, 13, 2949–2955. [Google Scholar] [CrossRef][Green Version]
View at Publisher | View at Google Scholar - Papait, A.; Vertua, E.; Magatti, M.; Ceccariglia, S.; De Munari, S.; Silini, A.R.; Sheleg, M.; Ofir, R.; Parolini, O. Mesenchymal Stromal Cells from Fetal and Maternal Placenta Possess Key Similarities and Differences: Potential Implications for Their Applications in Regenerative Medicine. Cells 2020, 9, 127. [Google Scholar] [CrossRef] [PubMed][Green Version]
View at Publisher | View at Google Scholar - Peng, S.-Y.; Wu, T.-H.; Lin, T.-Y.; Hii, L.-Y.; Chan, K.-S.; Fu, T.-Y.; Chang, S.-C.; Shen, P.-C.; Liu, K.-Y.; Shaw, S.W. Application of Cattle Placental Stem Cells for Treating Ovarian Follicular Cyst. World J. Stem Cells 2020, 12, 1366–1376. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Pérez-Merino, E.M.; Usón-Casaús, J.M.; Zaragoza-Bayle, C.; Duque-Carrasco, J.; Mariñas-Pardo, L.; Hermida-Prieto, M.; Barrera-Chacón, R.; Gualtieri, M. Safety and efficacy of allogeneic adipose tissue-derived mesenchymal stem cells for treatment of dogs with inflammatory bowel disease: Clinical and laboratory outcomes. Vet. J. 2015, 206, 385–390. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Peters, A.E.; Watts, A.E. Biopsy Needle Advancement during Bone Marrow Aspiration Increases Mesenchymal Stem Cell Concentration. Front. Vet. Sci. 2016, 3, 23. [Google Scholar] [CrossRef][Green Version]
View at Publisher | View at Google Scholar - Pittenger, M. F., Mackay, A. M., Beck, S. C., Jaiswal, R. K., Douglas, R., Mosca, J. D., et al. (1999). Multilineage potential of adult human mesenchymal stem cells. Science 284, 143–147. doi: 10.1126/science.284.5411.143.[PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Prządka, P.; Kiełbowicz, Z.; Osiński, B.; Dzimira, S.; Madej, J.A.; Nowacki, W.; Kubiak, K.; Reichert, P.; Cegielski, M. Reconstruction of cranial cruciate ligament in rabbits using polyester implants saturated with PRP, antlerogenic stem cells MIC-1 and their homogenate. Connect. Tissue Res. 2017, 58, 464–478. [Google Scholar] [CrossRef] [PubMed]
View at Publisher | View at Google Scholar - Rasmusson I., Uhlin M., Le Blanc K., Levitsky V. Mesenchymal stem cells fail to trigger effector functions of cytotoxic T lymphocytes. J. Leukoc. Biol. 2007;82:887–893. doi: 10.1189/jlb.0307140. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Rebelatto, C.K.; Aguiar, A.M.; Moretão, M.P.; Senegaglia, A.C.; Hansen, P.; Barchiki, F.; Oliveira, J.; Martins, J.; Kuligovski, C.; Mansur, F.; et al. Dissimilar Differentiation of Mesenchymal Stem Cells from Bone Marrow, Umbilical Cord Blood, and Adipose Tissue. Exp. Biol. Med. 2008, 233, 901–913. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Renzi, S.; Riccò, S.; Dotti, S.; Sesso, L.; Grolli, S.; Cornali, M.; Carlin, S.; Patruno, M.; Cinotti, S.; Ferrari, M. Autologous bone marrow mesenchymal stromal cells for regeneration of injured equine ligaments and tendons: A clinical report. Res. Vet. Sci. 2013, 95, 272–277. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Rhew, S.-Y.; Park, S.-M.; Li, Q.; An, J.-H.; Chae, H.-K.; Lee, J.-H.; Ahn, J.-O.; Song, W.-J.; Youn, H.-Y. Efficacy and safety of allogenic canine adipose tissue-derived mesenchymal stem cell therapy for insulin-dependent diabetes mellitus in four dogs: A pilot study. J. Vet. Med. Sci. 2021, 83, 592–600. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Rink B.E., Beyer T., French H.M., Watson E., Aurich C., Donadeu F.X. The fate of autologous endometrial mesenchymal stromal cells after application in the healthy equine uterus. Stem Cells Dev. 2018;27:1046–1052. doi: 10.1089/scd.2018.0056. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Saldinger L.K., Nelson S.G., Bellone R.R., Lassaline M., Mack M., Walker N.J., Borjesson D.L. Horses with equine recurrent uveitis have an activated CD4+ T-cell phenotype that can be modulated by mesenchymal stem cells in vitro. Vet. Ophthalmol. 2020;23:160–170. doi: 10.1111/vop.12704. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Sandersen C., Ceusters J., Fourez A., Tosi I., Graide H., Lejeune J.P., Serteyn D. Nerve stimulator-guided injection of autologous stem cells near the equine left recurrent laryngeal nerve. J. Vis. Exp. 2018;2018:58023. doi: 10.3791/58023. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Shah, K.; Drury, T.; Roic, I.; Hansen, P.; Malin, M.; Boyd, R.; Sumer, H.; Ferguson, R. Outcome of Allogeneic Adult Stem Cell Therapy in Dogs Suffering from Osteoarthritis and Other Joint Defects. Stem Cells Int. 2018, 2018, 7309201. [Google Scholar] [CrossRef][Green Version]
View at Publisher | View at Google Scholar - Shahid M.A., Kim W.H., Kweon O.K. Cryopreservation of heat-shocked canine adipose-derived mesenchymal stromal cells with 10% dimethyl sulfoxide and 40% serum results in better viability, proliferation, anti-oxidation, and in-vitro differentiation. Cryobiology. 2020;92:92–102. doi: 10.1016/j.cryobiol.2019.11.040. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Sharun, K.; Rawat, T.; Kumar, R.; Chandra, V.; Saxena, A.C.; Pawde, A.M.; Kinjavdekar, P.; Amarpal; Sharma, G.T. Clinical evaluation following the percutaneous transplantation of allogenic bone marrow-derived mesenchymal stem cells (aBM-MSC) in dogs affected by vertebral compression fracture. Vet. Anim. Sci. 2020, 10, 100152. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Smith, R.K.W.; Werling, N.J.; Dakin, S.G.; Alam, R.; Goodship, A.E.; Dudhia, J. Beneficial effects of autologous bone marrow-derived mesenchymal stem cells in naturally occurring tendinopathy. PLoS ONE 2013, 8, e75697. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Sole A., Spriet M., Galuppo L.D., Padgett K.A., Borjesson D.L., Wisner E.R., Brosnan R.J., Vidal M.A. Scintigraphic evaluation of intra-arterial and intravenous regional limb perfusion of allogeneic bone marrow-derived mesenchymal stem cells in the normal equine distal limb using 99mTc-HMPAO. Equine Vet. J. 2012;44:594–599. doi: 10.1111/j.2042-3306.2011.00530.x. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Soleimani, M., and Nadri, S. (2009). A protocol for isolation and culture of mesenchymal stem cells from mouse bone marrow. Nat. Protoc. 4, 102–106. doi: 10.1038/nprot.2008.221.[PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Sotiropoulou P.A., Perez S.A., Gritzapis A.D., Baxevanis C.N., Papamichail M. Interactions between human mesenchymal stem cells and natural killer cells. Stem Cells. 2006;24:74–85. doi: 10.1634/stemcells.2004-0359. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Spaas J.H., Broeckx S., Van De Walle G.R., Polettini M. The effects of equine peripheral blood stem cells on cutaneous wound healing: A clinical evaluation in four horses. Clin. Exp. Dermatol. 2013;38:280–284. doi: 10.1111/ced.12068. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Takehara Y., Yabuuchi A., Ezoe K., Kuroda T., Yamadera R., Sano C., Murata N., Aida T., Nakama K., Aono F., et al. The restorative effects of adipose-derived mesenchymal stem cells on damaged ovarian function. Lab. Investig. 2013;93:181–193. doi: 10.1038/labinvest.2012.167. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Textor J.A., Clark K.C., Walker N.J., Aristizobal F.A., Kol A., LeJeune S.S., Bledsoe A., Davidyan A., Gray S.N., Bohannon-Worsley L.K., et al. Allogeneic stem cells alter gene expression and improve healing of distal limb wounds in horses. Stem Cells Transl. Med. 2018;7:98–108. doi: 10.1002/sctm.17-0071. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - The Secretion Profile of Mesenchymal Stem Cells and Potential Applications in Treating Human Diseases|Signal Transduction and Targeted Therapy. Available online: https://www.nature.com/articles/s41392-022-00932-0 (accessed on 2 May 2022).
View at Publisher | View at Google Scholar - Ting, W.-J.; Shaw, S.W.; Hii, L.-Y.; Lin, T.-Y.; Chang, S.-C.; Liu, K.-Y.; Shen, P.-C.; Chen, T.-J.; Peng, S.-Y. Therapeutic Effects of Conditioned—DPBS from Amniotic Stem Cells on Lactating Cow Mastitis. Taiwan. J. Obstet. Gynecol. 2020, 59, 520–526. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Tremain, N., Korkko, J., Ibberson, D., Kopen, G. C., DiGirolamo, C., and Phinney, D. G. (2001). MicroSAGE analysis of 2,353 expressed genes in a single cell-derived colony of undifferentiated human mesenchymal stem cells reveals mRNAs of multiple cell lineages. Stem Cells 19, 408–418. doi: 10.1634/stemcells.19-5-408. [PubMed][CrossRef][Google Scholar]
View at Publisher | View at Google Scholar - Tripathy S. Cryopreservation of Mesenchymal Stem Cells (MSCs): Different approaches and applications. Int. J. Adv. Sci. Technol. 2017;1:435–456. [Google Scholar]
View at Publisher | View at Google Scholar - Van Vlasselaer, P., Falla, N., Snoeck, H., and Mathieu, E. (1994). Characterization and purification of osteogenic cells from murine bone marrow by two-color cell sorting using anti-Sca-1 monoclonal antibody and wheat germ agglutinin. Blood 84, 753–763.[PubMed] [Google Scholar]
View at Publisher | View at Google Scholar - Villagrán C.C., Schumacher J., Donnell R., Dhar M.S. A novel model for acute peripheral nerve injury in the horse and evaluation of the effect of mesenchymal stromal cells applied in situ on nerve regeneration: A preliminary study. Front. Vet. Sci. 2016;3:80. doi: 10.3389/fvets.2016.00080. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Villatoro, A.J.; Hermida-Prieto, M.; Fernández, V.; Fariñas, F.; Alcoholado, C.; Rodríguez-García, M.I.; Mariñas-Pardo, L.; Becerra, J. Allogeneic adipose-derived mesenchymal stem cell therapy in dogs with refractory atopic dermatitis: Clinical efficacy and safety. Vet. Rec. 2018, 183, 654. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Voga, M.; Adamic, N.; Vengust, M.; Majdic, G. Stem Cells in Veterinary Medicine—Current State and Treatment Options. Front. Vet. Sci. 2020, 7, 278. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Wang S., Yu L., Sun M., Mu S., Wang C., Wang D., Yao Y. The therapeutic potential of bone marrow mesenchymal stem cells in premature ovarian failure. Stem Cell Res. Ther. 2018;9 doi: 10.1186/s13287-018-1008-9. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Wang, Q. R., and Wolf, N. S. (1990). Dissecting the hematopoietic microenvironment. VIII. Clonal isolation and identification of cell types in murine CFU-F colonies by limiting dilution. Exp. Hematol. 18, 355–359.[PubMed][Google Scholar]
View at Publisher | View at Google Scholar - Wee, A. S., Lim, C. K., Merican, A. M., Ahmad, T. S., and Kamarul, T. (2013). Total cell pooling in vitro: an effective isolation method for bone marrow-derived multipotent stromal cells. In vitro Cell Dev. Biol. Anim. 49, 424–432. doi: 10.1007/s11626-013-9626-0.[PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Xiang Y., Zheng Q., Jia B., Huang G., Xie C., Pan J., Wang J. Ex vivo expansion, adipogenesis and neurogenesis of cryopreserved human bone marrow mesenchymal stem cells. Cell Biol. Int. 2007;31:444–450. doi: 10.1016/j.cellbi.2006.11.012. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Yamamoto, Y., Itoh, S., Yamauchi, Y., Matsushita, K., Ikeda, S., Naruse, H., et al. (2015). Density gradient centrifugation for the isolation of cells of multiple lineages. J. Cell Biochem. 116, 2709–2714. doi: 10.1002/jcb.25270. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Yong, K.W.; Wan Safwani, W.K.Z.; Xu, F.; Wan Abas, W.A.B.; Choi, J.R.; Pingguan-Murphy, B. Cryopreservation of Human Mesenchymal Stem Cells for Clinical Applications: Current Methods and Challenges. Biopreserv. Biobank. 2015, 13, 231–239. [Google Scholar] [CrossRef]
View at Publisher | View at Google Scholar - Yu, Y.; Valderrama, A.V.; Han, Z.; Uzan, G.; Naserian, S.; Oberlin, E. Human Fetal Liver MSCs Are More Effective than Adult Bone Marrow MSCs for Their Immunosuppressive, Immunomodulatory, and Foxp3+ T Reg Induction Capacity. Stem Cell Res. Ther. 2021, 12, 138. [Google Scholar] [CrossRef] [PubMed]
View at Publisher | View at Google Scholar - Zhu, H., Guo, Z. K., Jiang, X. X., Li, H., Wang, X. Y., Yao, H. Y., et al. (2010). A protocol for isolation and culture of mesenchymal stem cells from mouse compact bone. Nat. Protoc. 5, 550–560. doi: 10.1038/nprot.2009.238. [PubMed] [CrossRef] [Google Scholar]
View at Publisher | View at Google Scholar - Zuk, P.A.; Zhu, M.; Mizuno, H.; Huang, J.; Futrell, J.W.; Katz, A.J.; Benhaim, P.; Lorenz, H.P.; Hedrick, M.H. Multilineage cells from human adipose tissue: Implications for cell-based therapies. Tissue Eng. 2001, 7, 211–228. [Google Scholar] [CrossRef] [PubMed][Green Version]
View at Publisher | View at Google Scholar - Merlo, Barbara, Penelope Maria Gugole, and Eleonora Iacono.
View at Publisher | View at Google Scholar

 Clinic
Clinic
