Review Article | DOI: https://doi.org/10.31579/2835-8325/043
Morphological Organization of Precerebellar Nuclei and red Nucleus
Candidate of biological science, Assistant professor of pathophysiology department named D. A. Maslakov, Grodno State Medical University; Grodno State Medical University, 80 Gorky St,230009, Grodno, Belarus
*Corresponding Author: Elizaveta I Bon, Candidate of biological science, Assistant professor of pathophysiology department named D. A. Maslakov, Grodno State Medical University; Grodno State Medical University, 80 Gorky St,230009, Grodno, Belarus
Citation: Maksimovich N. Ye, Elizaveta I Bon, Lychkovskaya M.A, Golushko A.S, Pauliuchenkava D, (2023), Morphological Organization of Precerebellar Nuclei and red Nucleus, Clinical Research and Clinical Reports, 3(1); DOI:10.31579/2835-8325/043
Copyright: © 2023, Elizaveta I Bon. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: 27 September 2023 | Accepted: 17 October 2023 | Published: 08 November 2023
Keywords: truncus encephali nuclei; neurons; rats
Abstract
Objective. Analysis and systematization of literature data on the morphological organization of the truncus encephali nuclei.
Methods. The basis of this study was a review of the literature on this topic.
Results. The truncus encephali has a regulatory effect on many functions of the body simultaneously, both directly and through the centers of the spinal cord, and performs two main functions: reflex and conduction.
The reflex function is due to the presence in it of the nuclei of the cranial nerves, which are the centers of various reflex acts. The conduction function of the trunk is associated with the fact that there are ascending and descending pathways in the brain stem. Through the nerve fibers of these pathways, impulses are transmitted from the medulla spinalis to the encephalon and from the encephalon to the medulla spinalis.
Conclusion. The information presented in the review on the morphological organization of the rat truncus encephali nuclei provide a basis for further study of it in health and in cerebrovascular pathology, which will allow determining new ways of preventing and correcting these diseases.
Introduction
Most of the researches on the development of the cerebral cortex and the impact on this process of various experimental influences is carried out on laboratory rats. This determines the need for logical ideas about the structure of the brain in these animals [1,2,5].
Especially interesting is the study of the structures of the truncus encephali, which play an important role in the regulation of vital functions and act as a link and a center for switching information coming from various parts of the nervous system [22, 30].
The purpose of this review is to analyze and summarize the literature data on the morphological organization of the nuclei of the truncus encephali and their response to ischemia.
The truncus encephali includes the myelencephalon, the pons Varolii, and the mesencephalon. Sometimes referred to as the cerebellum and diencephalon (picture 1).
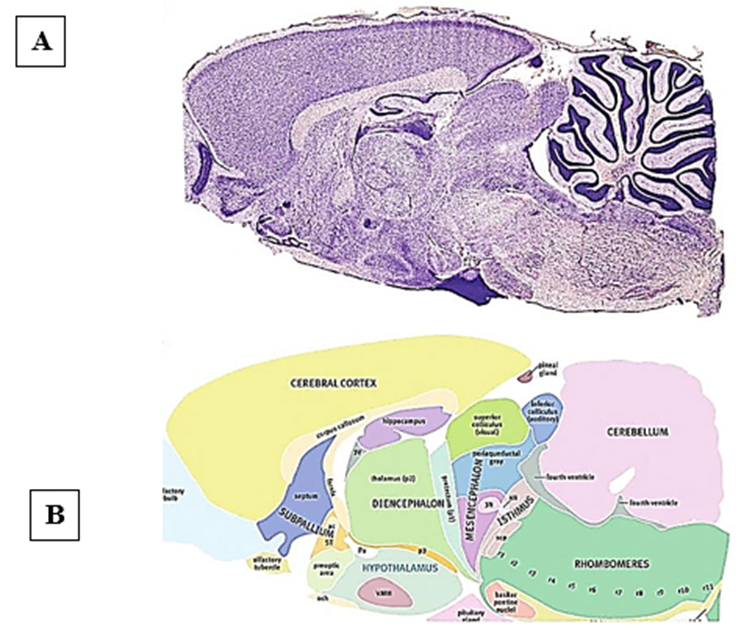
Picture 1. Rat brain
- - Nissl-stained photograph of the sagittal region of the rat brain. The incision was made towards the middle.
- - Picture color scheme [7].
This review will focus on the precerebellar truncus encephali nuclei and the red nucleus, which is a complex biological system for switching and integrating information coming from various parts of the spinal cord and brain.
The truncus encephali performs two main functions: reflex and conduction.
Reflex function is associated with the fact that the nuclei of cranial nerves and other clusters of nerve cells are laid here. Different nuclei are interconnected and functionally are the centers of various reflex acts.
Vital centers are located in this part of the brain. These include the centers of cardiac activity and the center of respiration. From the center, when it is excited, nerve impulses are transmitted along the motor nerves to the organ and cause a change in its activity (intensification or inhibition). So, the center of cardiac activity, located in the myelencephalon, sends impulses along the vagus nerve to the heart, exerting an inhibitory effect on it.
Together with the center of cardiac activity in the myelencephalon there is a vasomotor center, when excited, a change in the diameter of the blood vessels occurs. The medulla oblongata also contains the centers of many digestive and protective reflexes. These centers are connected with the corresponding organs by means of cranial nerves.
Truncus encephali exerts a regulatory influence simultaneously on many functions of the body, both directly and through the centers of the medulla spinalis. The conductive function of the Truncus encephali is due to the fact that they contain ascending and descending pathways. Along the nerve fibers of these pathways, impulses are transmitted from the medulla spinalis to the encephalon and from the encephalon to the medulla spinalis [18,22,29].
The nuclei of the pons Varolii
The basilar nuclei of the pons Varolii are clusters of small to medium-sized neurons located near the ventral surface of the mesencephalon.
Cytoarchitectonics
Neurons in the dorsal and medial regions of the basal ganglia are usually somewhat larger and have more dendrites compared to neurons in the ventral regions of the basal ganglia of the pons Varolii. Small neurons have an inhibitory effect and are GABAergic [16].
Myeloarchitectonics
Afferent pathways to the basilar nuclei of the pons Varolii begin from the 5th layer of neurons in the cerebral cortex of neurons. These are mainly sensorimotor, visual and temporal areas. In addition to the neocortex, afferents also form the cingulate gyrus, retrosplenial and agranular insular cortex. Innervation clusters tend to overlap [6,13].
Efferents coming from the cerebellum are represented by both glutamatergic and GABAergic fibers. Hypothalamic neurons form connections with the medial and dorsomedial part of the pons Varolii. These projections coincide with the afferent projections of the prefrontal cortex, playing an important role in the integration of the limbic and autonomic systems within the basal ganglia of the pons Varolii. In addition, the basal nuclei of the pons Varolii receive afferent fibers from many other spinal and stem centers [12,15].
Reticulopontine projections originate mainly from the ventral reticular, magnocellular and paramagnocellular nuclei. They are mainly GABAergic. The locus coeruleus and the suture nuclei form noradrenergic and serotonergic afferents.
Efferent pontocerebellar glutamatergic fibers terminate in mossy fibers in the granular layer of the cerebellar cortex [9,18].
The basal nuclei of the pons Varolii are not only the link between the cerebellar cortex and the neocortex, but can also be considered as the main integrating center. It is through the neurons of the pons Varolii that the actual visual, auditory and somatosensory information comes to the cerebellum. Perhaps the pons Varolii plays the role of a kind of interface that allows the cerebellum to process information coming from the neocortex [10,29].
The reticular nucleus of the pontine tegmentum is located dorsal to the basal nuclei of the pons Varolii; the fibers of its neurons are also involved in the formation of bryophyte fibers. Its cyto- and myeloarchitectonics is similar to the basal nuclei [8].
The reticulotegmental nucleus of the pons Varolii is located dorsal to the medial lemniscus. The central part of the nucleus consists of densely packed neurons, while in the pericentral nerve cells lie more diffusely [8,26].
Afferents come from the nuclei of the cerebellum, the cerebral cortex (cingulate and frontal), and a number of trunci encephali centers (optic tract nucleus, contralateral superior colliculus, medial terminal nucleus, lateral geniculate nucleus, anterior and posterior pretectal nuclei). Communication with the limbic system is maintained through the miliary nuclei of the hypothalamus [8,18,22].
Efferents are represented by both mossy fibers and projections ending in the nuclei of the hypoglossal nerve [8,16].
The lateral reticular nucleus is located in the myelencephalon ventrally, dorsolateral to the inferior olives, and ventromedial to the spinal tract of the trigeminal nerve. The lateral reticular nucleus plays an important role in the control of motor activity and coordination, as it is involved in the formation of mossy fibers. In addition, its neurons are part of the nociceptive system [19].
Cytoarchitectonics
It is magnocellular part is located dorsomedally and dorsolaterally, while its small cell part is located ventrally.
Myeloarchitectonics
The main afferent input is a bilateral medulla spinalis projection containing the ventral flexor reflex tract, the ipsilateral forelimb tract, and propriospinal neuron fibers. This indicates the important role of the lateral reticular nucleus in the integration and coordination of motor activity. In addition, the lateral reticular nucleus receives afferents from the contralateral red nucleus, the medial nucleus of the cerebellum, the thalamus, the hypothalamus, and the cerebral cortex [12,13].
The lateral reticular nucleus is involved in the formation of mossy fibers to the cerebellar cortex and to the vermis [15]. In addition, it sends efferents to the lower olives and the medulla spinalis.
Neuroanatomical and electrophysiological studies have shown that the neurons of the lateral reticular nucleus are involved in the control of autonomic cardiovascular activity, respiratory activity, and pain perception [8,16,22].
Olives inferioris
The inferior olives are a group of nuclei located ventromedially in the caudal part of the myelencephalon.
Cytoarchitectonics
The neurons of the nuclei of the olives inferioris of the rat are small and have four to seven primary dendrites. There are two types of olivar neurons: globular (their dendrites curve in the form of arches towards the perikaryon) and simple (their dendrites branch out from the perikaryon). Olive neurons are interconnected not only by synapses, but by gap junctions found in the spiny apparatus of their dendrites [23]. Neurotransmitters are GABA, enkephalin, substance P, cholecystokinin [28].
Myeloarchitectonics
The olives inferioris receive afferents carrying cutaneous tactile and nociceptive information from the spinal cord and trigeminal nuclei. Afferent GABAergic fibers come from the lateral mesencephalic nucleus, the nucleus of the pretectal region, the reticular formation, the cerebellum, the nucleus of the optic tract, and the accessory optic nuclei. Connections of the olives inferioris with the red nucleus, sensorimotor and parietal cortex have also been described [27].
Efferent connections
The olive inferior neurons are the only source of ascending cerebellar climbing fibers terminating in the dendritic tree of Purkinje cells. The neurotransmitters in these fibers are glutamate, enkephalin, and cholecystokinin [28].
The nuclei of the inferior olives play an important role in the regulation of motor activity, the autonomic system, and modulation of the cerebellum [23].
Red nucleus
The red nucleus of the rat is approximately ovoid in shape and is located bilaterally in the tegmentum of the midbrain.
Cytoarchitectonics.
In the red nucleus, four types of neurons are distinguished according to the size of the perikaryon.
In the caudal part of the nucleus, there are giant (perikaryon diameter more than 40 micrometers) and large (perikaryon diameter 26-40 micrometers) neurons, and in the rostral part - medium (perikaryon diameter 20-25 micrometers) and small neurons (<20>
According to the chromatophilia of the cytoplasm, three types of neurons are distinguished: hyperchromic, hypochromic and achromatic. Hyperchromic ones are mainly localized in the caudal region, hypochromic ones in the rostral region, and achromatic ones are diffusely located throughout the nucleus. In addition, there are small- and large-nuclear neurons in the red nucleus [11].
Myeloarchitectonics
The red nucleus forms afferent connections with the nuclei of the cerebellum, thalamus, as well as with the neurons of the fifth layer of the agranular frontal cortex.
Neurotransmitters are aspartate and GABA [21].
The magnocellular caudal part of the red nucleus gives rise to efferent rubrospinal and rubrobulbar tracts. Rubrospinal fibers switch on the nuclei of the reticular formation, the nuclei of the trigeminal and facial nerves. The small-celled rostral part gives rise to the central tegmental tract, which descends to the ipsilateral olives inferioris, and also forms efferent connections with the thalamus. In addition, the red nucleus gives rise to the rubrocerebellar pathway [20,25].
Efferent pathways use glutamate and aspartate as neurotransmitters [24,25].
It is believed that large neurons of the red nucleus are involved in the implementation of motor functions and maintain muscle tone, while small neurons are part of the antinociceptive system [20].
The information presented in the review on the morphological organization of the rat truncus encephali nuclei provide a basis for further study of it in health and in cerebrovascular pathology, which will allow determining new ways of preventing and correcting these diseases.
References
- Antonova O.G, Khazov P.D. (2004). Dynamics of ischemic strokes of the truncus encephali during MRI // Rossiyskiy mediko-biologicheskiy vestnik im. akad. I.P.Pavlova. Russian medical and biological bulletin named after acad. I.P. Pavlova, 108-112.
View at Publisher | View at Google Scholar - Bon, E I. (2023). Classical Methods for Studying the Structure of Cells / E.I. Bon, N. Ye. Maksimovich, S. M. Zimatkin, N.V. Kokhan // Biomed J Sci & Tech Res, 50(5):1-2.
View at Publisher | View at Google Scholar - Konovalov A.N, Kornienko V.N, Pronin I.N. (2001). Hematomas and hidden vascular malformations of the brainstem // Meditsinskaya vizualizatsiya. Medical imaging, 15-21.
View at Publisher | View at Google Scholar - Naumov N.G, Oleinik E.A, Droblenkov A.V. (2016). Changes in astrocytes after blood flow restriction in the nucleus accumbens in rats // Morfologiya. Morphology, 37-42.
View at Publisher | View at Google Scholar - Naumov N.G. (2015). Postischemic changes in brain neurons in the area of limited blood flow and the effectiveness of their correction with Metaprot in rats // Zhurnal anatomii i gistopatologii. Journal of Anatomy and Histopathology, 88-112.
View at Publisher | View at Google Scholar - Naumov N.G. (2016). Reactive changes in astrocytes of the nucleus accumbens of the forebrain after blood flow restriction in the basin of both common carotid arteries in rats // Morfologiya. Morphology, 22-27.
View at Publisher | View at Google Scholar - Center for Medical Information [Electronic resource]. - Access mode: https://cmi.to/. – Access date: 23.07.2021.
View at Publisher | View at Google Scholar - Alstermark B., Ekerot C-F. (2013). The lateral reticular nucleus: a precerebellar centre providing the cerebellum with overview and integration of motor functions at systems level. A new hypothesis // The Journal of Physiology, 5453-5458.
View at Publisher | View at Google Scholar - Balaban C.D, Beryozkin G. Organization of vestibular nucleus projections to the caudal dorsal cap of kooy in rabbits // Journal of Neuroscience, 1217-1236.
View at Publisher | View at Google Scholar - Behnke V.K, Stevenson M.E. (2016). Inactivation of the cerebellar fastigial nuclei alters social behavior in the rat // Journal of Behavioral Neuroscience, 552-560.
View at Publisher | View at Google Scholar - Benjamin E. A, Dorothy E. O. Absolute number of parvicellular and magnocellular neurons in the red nucleus of the rat midbrain: a stereological study // Journal of Anatomy, 406-415.
View at Publisher | View at Google Scholar - Bin L, Zhuang Q-X, Gao H-R. Medial cerebellar nucleus projects to feeding-related neurons in the ventromedial hypothalamic nucleus in rats// Journal of Brain Structure and Function, 957-951.
View at Publisher | View at Google Scholar - Bosco G, Casabona A, Giaquinta G. (1996). Influences exerted by the frontal eye field on accessory oculomotor nuclei neurons of the rat // Journal of Archives Italiennes de Biologie, 305-316.
View at Publisher | View at Google Scholar - Caltana, L, Merelli A, Lazarowski A, Brusco A. (2009). Neuronal and glial alterations due to focal cortical hypoxia induced by direct cobalt chloride (CoCl2) brain injection // Journal of Neurotoxicity Research, 348-358.
View at Publisher | View at Google Scholar - Courville J, Diakiw N. (1976). Cerebellar corticonuclear projection in the cat. The vermis of the anterior and posterior lobes // Journal of Brain Research, 1-20.
View at Publisher | View at Google Scholar - Federico C, Serapide M.F, Parenti R. (2005). The basilar pontine nuclei and the nucleus reticularis tegmenti pontis subserve distinct cerebrocerebellar pathways // Journal of Progress in Brain Research, 44-49.
View at Publisher | View at Google Scholar - Kofler, J. (2011). Microglia: key innate immune cells of the brain // Journal of Toxicologic Pathology, 103-114.
View at Publisher | View at Google Scholar - Mohammad S.B, Yuanjun L. (2019). Divergent projections of single pontocerebellar axons to multiple cerebellar lobules in the mouse // The Journal of Comparative Neurology, 1966-1985.
View at Publisher | View at Google Scholar - Nakamura H. (2018). Cerebellar projections to the ventral lateral geniculate nucleus and the thalamic reticular nucleus in the cat // Journal of Neuroscience Research, 63-74.
View at Publisher | View at Google Scholar - Olyntho-Tokunaga H, Pinto M.L, Souccar C. (2008). Projections from the anterior interposed nucleus to the red nucleus diminish with age in the mouse // Journal of Anatomia, Histologia, Embryologia, 438-441.
View at Publisher | View at Google Scholar - Orçun O.O, Xiaolu W, Binda F. (2020). Differential Coding Strategies in Glutamatergic and GABAergic Neurons in the Medial Cerebellar Nucleus // The Journal of Neuroscience, 159-170.
View at Publisher | View at Google Scholar - Paxinos G. (2004). The Rat Nervous System // Journal of Elsevier Academic Press, 1780.
View at Publisher | View at Google Scholar - Porzionato A, Macchi V, Stecco C. (2008). Morphometric analysis of the inferior olivary complex in infants and adults // Italian Journal of Anatomy and Embryology. – 2008. – N2. – P. 65-73.
View at Publisher | View at Google Scholar - Rácz E, Gaál B, Matesz C. (2016). Heterogeneous expression of extracellular matrix molecules in the red nucleus of the rat // Journal of Neuroscience, 1-17.
View at Publisher | View at Google Scholar - Roger N. L. (2016). Cortical projections to the red nucleus and the brain stem in the rhesus monkey // Journal of Brain Research, 15:28-30.
View at Publisher | View at Google Scholar - Ruben R-M, Uğur T. (2017). The Medial and Lateral Lemnisci: Anatomically Adjoined but Functionally Distinct Fiber Tracts // Journal of World Neurosurgery, 3:241-250.
View at Publisher | View at Google Scholar - Yamaguchi K. (2019). Development of the human principal inferior olivary nucleus: A morphometric and computerized 3D-reconstruction study // Journal of Neuroscience Letters, 8:1002-1031.
View at Publisher | View at Google Scholar - You Y, Yuhong F, Charles W. (2014). The inferior olive of the C57BL/6J mouse: a chemoarchitectonic study // Journal of The Anatomical Record, 2:289-300.
View at Publisher | View at Google Scholar - Zhang H, Gamlin P.D. (1998). Neurons in the posterior interposed nucleus of the cerebellum related to vergence and accommodation. I. Steady-state characteristics // Journal of Neurophysiology, 3:1255-1269.
View at Publisher | View at Google Scholar - Maksimovich, N. Ye. (2020). Rat brain and it is reaction to ischemia: monograph // N. Ye. Maksimovich, E. I. Bon, S. M. Zimatkin. – Grodno: GrSMU, 17:240
View at Publisher | View at Google Scholar

 Clinic
Clinic
