Research Article | DOI: https://doi.org/10.31579/2835-7957/027
Effect of Adenosine Receptor Modulation on Caffeine-Induced Motor Activity in Rats
1 Department of Basic Medical Sciences, Faculty of Medicine, Yarmouk University, Irbid-Jordan.
2 Department of Pharmacology, Faculty of Medicine, Assiut University, Egypt.
3 M.B.B. Ch, Faculty of Medicine, Assiut University, Egypt.
*Corresponding Author: Romany H. Thabet, Department of Basic Medical Sciences, Faculty of Medicine, Yarmouk University, Irbid-Jordan.
Citation: Romany H. Thabet, Youssef Romany Helmy, (2023), Effect of Adenosine Receptor Modulation on Caffeine-Induced Motor Activity in Rats, Clinical Reviews and Case Reports, 2(6); DOI:10.31579/2835-7957/027
Copyright: © 2023, Romany H. Thabet. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: 16 November 2023 | Accepted: 01 December 2023 | Published: 08 December 2023
Keywords: adenosine; a1 receptors; a2 receptors; caffeine; locomotor activity; forced performance
Abstract
Controversies regarding benefits and risks of coffee consumption still exist, but the limitless health-promoting benefits of coffee outclass its few reported toxic effects. This study is devoted to the investigation of a potential inhibitory effect of adenosine and its analogs on the enhanced motor activity induced by caffeine. This was carried out by studying the effect of pretreatment of rats with adenosine and its analogs on caffeine enhanced spontaneous coordinate locomotor activity and forced motor performance in rats. Adenosine elicited a rapid inhibitory effect on spontaneous motor activity and forced performance of rats. N6-cylcopentyl adenosine (CPA), an A1 agonist, exerted a long-lasting inhibitory effect on motor activity while 5'-(N-Cyclopropyl) carboxamidoadenosine (CPCA), an A2 agonist, didn’t cause any change in motor activity. On the contrary, caffeine produced profound central nervous system (CNS) excitability, restlessness and marked increase of motor activity of rats. This stimulant effect was opposed by pretreating rats with adenosine and CPA but not with CPCA. In conclusion, the current study implies that A1 receptors are involved in the control of caffeine-induced motor activity while excluding any role of A2 receptors.
Introduction
Coffee confers many diverse health effects; some are beneficial and others are deleterious. It contains not only caffeine but other bioactive polyphenolic compounds. Intake of moderate doses of caffeine may be beneficial for cardiovascular conditions1. On the other hand, patients with irregular hypertension should avoid large doses of caffeine 2. There was a study bring to light that caffeine is a double-edged sword on blood pressure regulation; moderate coffee consumption may reduce the risk of developing arterial hypertension however in some cases it has hypertensinogenic effects3. Caffeine is useful in reducing the causes of death from heart disease in females and those with regular blood pressure, while this did not appear in males and those with irregular blood pressure 4. Caffeine intake delay parasympathetic pacemaker after exercise so recovery of heart rate and blood pressure take longer time 5. On the same side, an effect is shown that caffeine is stimulating sympathetic cardiac control and delaying parasympathetic reactivation after exercise6. Energy drinks mediated by a positive inotropic effect, which increase the rate, output and contractility of heart 7. It is believed that caffeinated energy drink causes development of coronary event8. A study suggests a positive relationship between caffeine and the antioxidant capacity of red blood cells 9. Caffeine reduces acute renal insufficiency and, in this sense, protects the heart so there is an important aspect to cardiorenal protective effects of coffee consumption10. Caffeine is a natural stimulant of the CNS and has psychotropic properties11,12. By inhibiting adenosine's ability to connect to the adenosine A1 receptor, caffeine increases the release of the neurotransmitter acetylcholine13,14. Similar to adenosine, caffeine has a three-dimensional structure that enables it to attach to and inhibit its receptors15,16. Low to moderate doses of caffeine may help treat some neurological conditions like symptoms of attention deficit hyperactivity disorder and major depressive disorder in humans, as well as boost alertness, energy, and concentration17,18. According to studies, regular coffee drinkers are also 45 percent less likely to commit suicide and have a lower risk of developing dementia, Parkinson’s disease and Alzheimer's disease19-22 . Caffeine views coffee as a healthy beverage, but like other things, an excessive amount can have negative consequences. Excessive consumption of caffeine can lead to caffeine intoxication (caffeinism), which includes negative symptoms such as anxiety, agitation, insomnia, gastrointestinal disturbances, tremors, mental disturbances, confusion, and hallucinations23. Moreover, depending on the sensitivity, in rare cases, it can also cause death24. Transient coffee consumption increases the risk of stroke, especially among irregular drinker’s caffeine decreases cerebral blood flow leads to damage and eventually kills brain cells, known as subcortical vascular dementia or small vessel disease25-27.Caffeine excites CNS in both human and animal models; it rapidly penetrates the blood-brain barrier and it is a competitive inhibitor of adenosine receptors in the brain, specifically A1 and A2A28,29. The A1 receptors are located in high levels in the cerebellum, cerebral cortex, hippocampus, and hypothalamus, whereas A2 receptors are found largely in the olfactory bulb, striatum, and nucleus accumbens (NAs). 30
The current study aimed at investigating the effect of pretreating rats with adenosine receptor A1 and A2 receptors on caffeine-induced locomotor and forced performance in rats. This study provides an insight on the importance of how modulation of adenosine receptors might affect the neurological actions of caffeine and consequently might clarify a possible mechanistic approach to caffeine.
Materials and methods Materials
Materials
Chemicals:
The following chemicals were used and obtained from the sources indicated:
1.Adenosine (ADO) (ICN biomedicals, Inc). ADO is fairly soluble in cold water, soluble in room temperature water, freely soluble in hot water, soluble in I N hydrochloric acid (50 mg/ml – clear and colorless), can be suspended in 8% tween 20 and insoluble in alcohol.
2.N6-cylcopentyl adenosine (CPA) (A1 agonist) (ICN biomedicals, Inc). CPA is soluble in ethanol and moderately soluble in water.
3.5'-(N-Cyclopropyl) carboxamidoadenosine (CPCA) (A2 agonist) (ICN biomedicals, Inc). CPCA is moderately soluble in ethanol, slightly soluble in warm water, very soluble in dilute aqueous acid and can be suspended in 8% tween 20.
4.Caffeine (ICN biomedicals, Inc). Caffeine is moderately soluble in water at room temperature (2 g/100 mL) and also moderately soluble in ethanol (1.5 g/100 mL).
5.Pentylenetetrazole (PTZ) (Sigma, USA). PTZ is soluble in normal saline and water.
All drugs were used as freshly prepared solutions in distilled water except CPA which was dissolved in 8% ethanol and CPCA was suspended in 8% tween 20.
Animals:
Adult male rats weighing 150-200g were used. The animals were group housed in plastic cages and maintained under standard laboratory conditions with a natural light-dark cycle. Rats were left to acclimatize to the environment for at least a week before the experiments. Food and water were allowed ad libitum.
Effect of adenosine and its analogs on motor activity of rats:
5 groups of rats each was comprised of 5 animals.
Treatment schedules:
Group A: was given i.p 0.5 ml of 8% tween 20.
Group B: was given i.p 0.5 ml of 8% ethanol.
Group C: was given adenosine i.p in a dose of 100 mg/kg.
Group D: was given CPA i.p in a dose of 10 mg/kg.
Group E: was given CPCA i.p in a dose of 10 mg/kg.
The motor activity was determined by:
1.Activity cages: (For screening of locomotor activity)
Rats were placed inside an acrylic transparent cage that rests on a sensor platform. It detects ambulatory movements as well as stereotypic activity like grooming, scratching, digging, etc. Vibrations caused by the animal activity produce proportional electrical signals. These are electrically processed to generate trigger pulses and drive a digital counter. Every count registered is accompanied by a flash. Activity recording was continued for 180 minutes. Activity records were taken for 1 minute each at 1, 5, 30, 60, 120 and 180 minutes after giving the drugs mentioned above.31
2.Rotarod test: (For screening of forced motor performance) Rats were allowed to remain on a rotating rod until falling off. The length of time the rat remained on the rod was recorded. The falling latency was recorded for each group at 1, 5, 15, 30, 60, 120 and 180 minutes after giving the drug.32
Effect of adenosine receptor modulation on motor activity of rats induced by caffeine:
7 groups of rats each was comprised of 5 animals
Treatment schedules:
Group A: was given caffeine in therapeutic doses (100 mg/kg).
Group B: was given 0.5 ml of 8% tween 20.
Group C: was given ADO 100 mg/kg 5 minutes before caffeine 50 mg/kg.
Group D: was given 0.5 ml of 8% ethanol.
Group E: was given CPA 10 mg/kg 60 minutes before caffeine 50 mg/kg.
Group F: was given CPCA 10 mg/kg 60 minutes before caffeine 50 mg/kg. The motor activity of each group was determined as discussed above.
Results
Effect of adenosine and its analogs on spontaneous motor activity of rats:
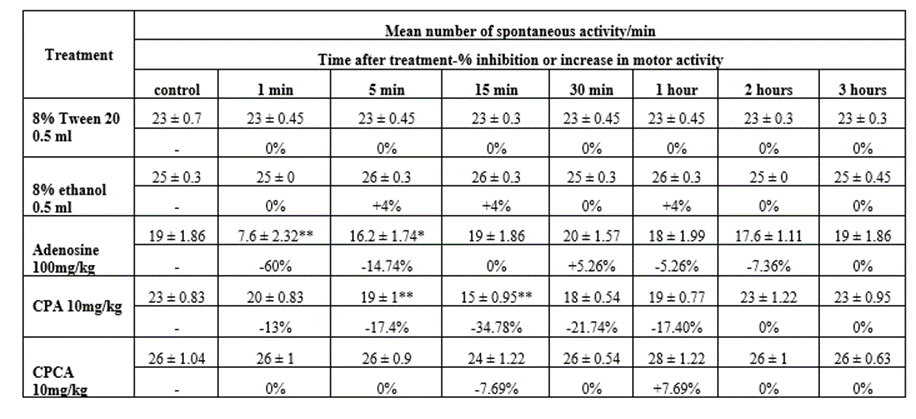
A) Effect of i.p injection of 8% Tween 20 and 8% ethanol on the spontaneous coordinate activity in rats (Table 1):
Intraperitoneal injection of o.5 ml of each of 8% Tween 20 and 8% ethanol showed no significant changes in the spontaneous coordinate activity of rats.
B) Effect of adenosine on the spontaneous coordinate activity in rats (Table 1):
Intraperitoneal injection of adenosine in a dose of 100 mg/kg produced significant decrease in spontaneous coordinate activity of rats. Maximal effect was noticed 1 minute after administration. The decrease of spontaneous activity 5 minutes after administration was less. Spontaneous activity returned to normal 15 minutes after administration.
C) Effect of CPA on the spontaneous coordinate activity in rats (Table 1):
Intraperitoneal injection of N6-cyclopentyladenosine (CPA) in a dose of 10 mg/kg produced also a decrease in the spontaneous motor activity of rats. The effect was maximal 15 minutes after administration, and then motor activity returned to normal 2 hours post-injection.
D) Effect of CPCA on the spontaneous coordinate activity in rats (Table 1):
N.B: Data represent mean ± SE of 5 observations
* P< 0>
+ % = percentage increase of motor activity
- % = percentage inhibition of motor activity
Intraperitoneal injection of CPCA in a dose of 10 mg/kg showed no significant changes in the spontaneous activity of rats.
Table 1: Effect of I.P. injection of 8% Tween 20, 8% ethanol, Adenosine, N6-cyclopentyladenosine (CPA), and 5-(N-cyclopropyl) carboxamidoadenosine (CPCA) each alone on spontaneous coordinate activity in rats.
Effect of caffeine after adenosine receptor modulation on spontaneous motor activity of rats:
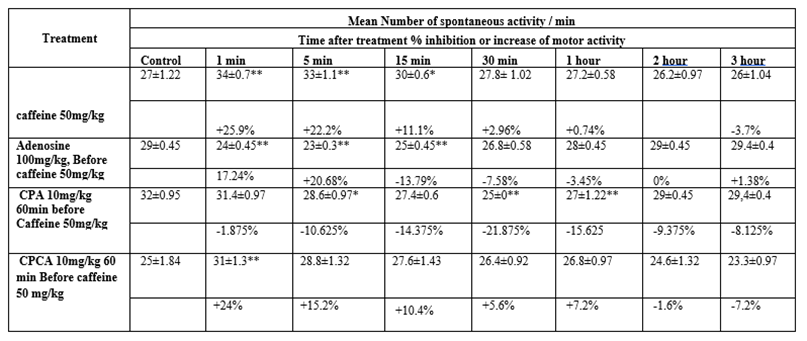
A) Effect of caffeine on the spontaneous coordinate activity in rats (Table 2):
Intraperitoneal injection of caffeine in a dose of 50 mg/kg in rats showed increase in spontaneous activity. The effect was maximal 1 minute after injection. This increase of motor activity continued up to one-hour post-injection.
B) Effect of caffeine after administration of adenosine on the spontaneous coordinate activity of rats (Table 2):
Intraperitoneal injection of adenosine in a dose of 100 mg/kg 5 minutes before caffeine in a dose of 50 mg/kg produced inhibition of spontaneous activity. The maximal inhibition was achieved 5 minutes after caffeine, then returned to normal two hours post-injection.
C) Effect of caffeine after administration of CPA on the spontaneous coordinate activity of rats (Table 2):
Intraperitoneal injection of CPA in a dose of 10 mg/kg 60 minutes before caffeine in a dose of 50 mg/kg produced inhibition of spontaneous activity. The maximal inhibition was recorded 30 minutes after caffeine injection. Then, motor activity returned to normal 3 hours post-injection.
D) Effect of caffeine after administration of CPCA on the spontaneous coordinate activity of rats (Table 2):
N.B: Data represent mean ±SE of 5 observations
*P <0>
+%=percentage increase of motor activity
-%= percentage inhibition of motor activity
Intraperitoneal injection of CPCA in a dose of 10 mg/kg 60 minutes before caffeine in a dose of 50 mg/kg, does not affect the action of caffeine. That means that caffeine produced the same significant increase of the spontaneous activity, starting 1 minute after its injection and continuing for 1 hour, as when injected alone.
Effect of adenosine and its analogs on forced motor performance of rats “rotarod test”:
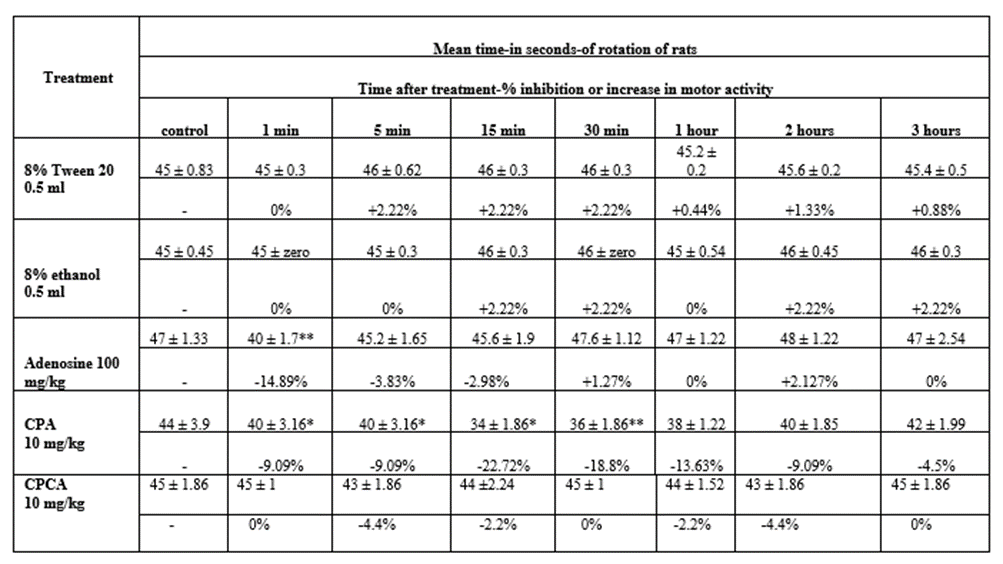
A) Effect of i.p injection of 8% Tween 20 and 8% ethanol on the forced motor performance of rats (Table 3):
Intraperitonal injection of each of 8% Tween 20 and 8% ethanol produced no changes in the forced motor performance of rats.
B) Effect of adenosine on the forced motor performance of rats (Table 3):
Intraperitoneal injection of adenosine in a dose of 100 mg/kg produced inhibition of the forced motor performance of rats. The maximal inhibition was 1 minute after injection.
C) Effect of CPA on the forced motor performance of rats (Table 3):
Intraperitoneal injection of CPA in a dose of 10 mg/kg produced inhibition of the forced motor performance. The inhibition started 1 minute after injection. Maximum inhibition was 15 minutes after injection.
D) Effect of CPCA on the forced motor performance of rats (Table 3)
N.B: Data represent mean ± SE of 5 observations
* P< 0>
+ % = percentage increase of motor activity
- % = percentage inhibition of motor activity
Intraperitoneal injection of CPCA in a dose of 10 mg/kg produced no significant changes in the forced motor performance in rats.
Effect of caffeine after adenosine receptor modulation on the forced motor performance of rats:
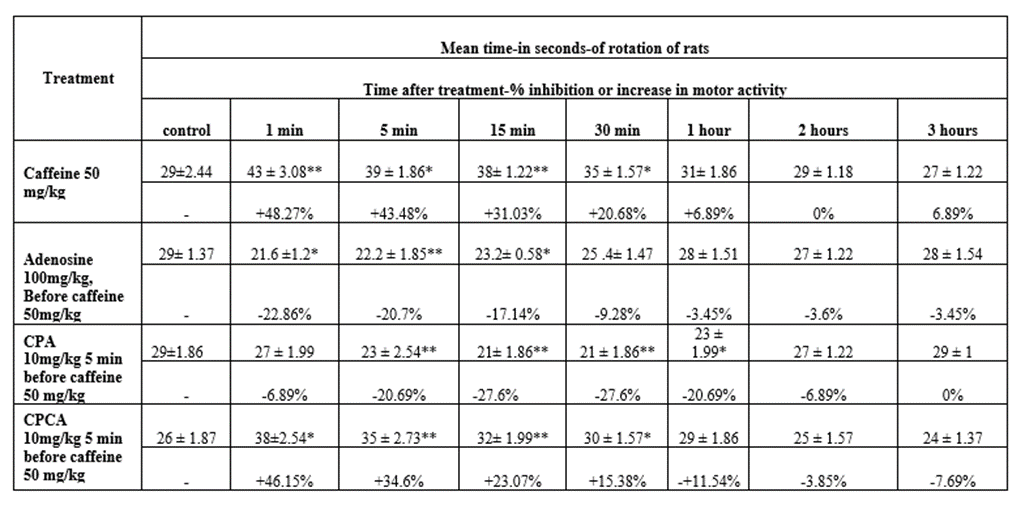
A) Effect of caffeine on the forced motor performance in rats (Table 4):
Intraperitoneal injection of caffeine in a dose of 50 mg/kg showed increase in the forced motor performance of rats. The effect was maximal 1 minute after injection and continued for 2 hours after injection.
B) Effect of caffeine on the forced motor performance of rats after administration of adenosine (Table 4):
Intraperitoneal injection of adenosine 100 mg/kg 5 minutes before caffeine in a dose of 50 mg/kg, produced inhibition of the forced motor performance. This inhibition was maximal 1 minute after injection of caffeine then the motor performance returned to normal 1-hour post-injection.
C) Effect of caffeine on the forced motor performance of rats after administration of CPA (Table 4):
Intraperitoneal injection of N6-cyclopentyladenosine in a dose of 10 mg/kg 60 minutes before caffeine in a dose of 50 mg/kg produced inhibition of forced motor performance of rats. This inhibition was maximal 30 minutes after injection of caffeine, then motor performance returned into normal 3 hours post-injection.
D) Effect of caffeine on the forced motor performance of rats after administration of CPCA (Table 4):
* P< 0>
+ % = percentage increase of motor activity
- % = percentage inhibition of motor activity
Intraperitoneal injection of CPCA in a dose of 10 mg/kg 60 minutes before caffeine 50 mg/kg does not affect significantly the forced motor performance induced by caffeine. This means that caffeine showed the same increase in the motor performance, which reached its maximal 1 minute after caffeine injection. But it was observed that the increase in motor performance lasted for 1 hour only, instead of 3 hours as when it was administered alone.
Discussion
Adenosine elicited a rapid inhibitory effect on spontaneous motor activity and forced performance of rats. CPA, an A1 agonist, exerted a long-lasting inhibitory effect on motor activity while CPCA, an A2 agonist, didn’t cause any change in motor activity. On the contrary, caffeine, a CNS stimulant, produced profound CNS excitability, restlessness and marked increase of motor activity of rats. This stimulant effect was opposed by pretreating rats with adenosine and CPA but not with CPCA. The above results indicate that the CNS depressant activity of adenosine imply that A1 receptors are involved in the control of motor activity while excluding any role of A2 receptors. These results are in agreement with those observed with other investigators, Popoli et al. 33 have demonstrated that adenosine A1 antagonists stimulate motor activity and Latini and his colleagues 34 concluded that adenosine is an endogenous neuromodulator that exerts its depressant effects on neurons by acting on the A1 receptor subtype. In addition, a study by Marston et al. 35 showed that adenosine A1 agonist, CPA reduced spontaneous motor activity and that CPA-induced locomotor depression was attenuated by adenosine A1 receptor selective antagonists such as DPCPX, FK 453 and FK 352 but not by the A2 receptor antagonist KF 17837.On the contrary, caffeine, a CNS stimulant produced profound CNS excitatory activity and caused restlessness and marked increase of spontaneous and forced motor activity of rats. This stimulant effect of caffeine was opposed by pretreating rats with adenosine and CPA (A1 agonist) while it was not changed after pretreatment with CPCA (A2 agonist). These results suggest a possible antagonistic effect of caffeine on adenosine A1 receptors.Caffeine's effect on motor function was observed to be strongly dose-dependent; it exhibits biphasic effects, with low doses that increase motor function (mediated by the A2A receptor) and high doses that decrease it (mediated by the A1 receptor).36 On NAs, caffeine has been shown to predominately raise extracellular levels of dopamine and glutamate which are linked to
Slocomotor stimulating effects.37 In the basal ganglia, caffeine may affect locomotor activity, too. Since A1 and A2A receptors are expressed in the striatum, A1 receptors can be detected in both direct pathway spiny projection neurons (dSPNs) and indirect pathway spiny projection neurons (iSNP) while A2A receptors are only found in iSPNs. Dopamine D2-receptors have also been detected in iSPNs, and the interactions between D2 and A2A receptors have received attention.38 Caffeine, at larger dosages, primarily targets A2A-D2 heteromers and inhibits A2A receptors, resulting in an increase in dopamine activity in the D2 pathway, which eventually improves thalamic inhibition and decrease movement.39 However, at lower doses, caffeine blocked the striatal adenosine receptor, leading to a decrease in the production of nerve growth factor (NGF) immediate early gene (NGFI-A) and NGFI-B mRNA and hence, a stimulating effect.40
In skeletal muscle, the locomotor improvement might be related to modulating calcium homeostasis in muscle fibers and enhancing the sensitivity of myofilaments to calcium ions31. An animal study discovered structural changes in the stained sections of caffeine-treated mice. In this study, increased dendritic length was related to enhanced motor performance; hence, coffee consumption appears to play a favorable effect in the continuous maintenance of cortical neurons in a dose-dependent way.41 However, higher amounts of caffeine have been demonstrated to inhibit calcium absorption and storage in the sarcoplasmic reticulum of striated muscle, as well as to promote Ca++ translocation across the plasma membrane.42
Conclusion
Adenosine elicited a rapid inhibitory effect on spontaneous motor activity and forced performance of rats. CPA, an A1 agonist, exerted a long-lasting inhibitory effect on motor activity while CPCA, an A2 agonist, didn’t cause any change in motor activity. On the contrary, caffeine, a CNS stimulant, produced profound CNS excitability, restlessness and marked increase of motor activity of rats. This stimulant effect was opposed by pretreating rats with adenosine and CPA but not with CPCA. The above results indicate that the CNS depressant activity of adenosine imply that A1 receptors are involved in the control of motor activity while excluding any role of A2 receptors.
References
- Voskoboinik A, Koh Y, Kistler PM. (2019). Cardiovascular effects of caffeinated beverages. Trends in cardiovascular medicine.29(6):345-350.
View at Publisher | View at Google Scholar - Rodríguez-Artalejo F, López-García E. (2018). Coffee Consumption and Cardiovascular Disease: A Condensed Review of Epidemiological Evidence and Mechanisms. Journal of agricultural and food chemistry. 66(21):5257-5263.
View at Publisher | View at Google Scholar - Surma S, Oparil S. (2021). Coffee and Arterial Hypertension. Current hypertension reports. 23(7):38.
View at Publisher | View at Google Scholar - Chen S, Li J, Gao M, et al. (2022). Association of caffeine intake with all-cause and cardiovascular mortality in elderly patients with hypertension. Frontiers in nutrition. 9:1023345.
View at Publisher | View at Google Scholar - Benjamim CJR, Monteiro LRL, Pontes YMM, et al. (2021). Caffeine slows heart rate autonomic recovery following strength exercise in healthy subjects. Revista portuguesa de cardiologia.40(6):399-406.
View at Publisher | View at Google Scholar - Benjamim CJR, Kliszczewicz B, Garner DM, et al. (2020). Is Caffeine Recommended Before Exercise? A Systematic Review To Investigate Its Impact On Cardiac Autonomic Control Via Heart Rate And Its Variability. Journal of the American College of Nutrition.39(6):563-573.
View at Publisher | View at Google Scholar - Lippi G, Cervellin G, Sanchis-Gomar F. (2016). Energy Drinks and Myocardial Ischemia: A Review of Case Reports. Cardiovascular toxicology.16(3):207-212.
View at Publisher | View at Google Scholar - Scott MJ, El-Hassan M, Khan AA. (2011). Myocardial infarction in a young adult following the consumption of a caffeinated energy drink. BMJ case reports. 2011.
View at Publisher | View at Google Scholar - D'Alessandro A, Fu X, Reisz JA, et al. (2020). Stored RBC metabolism as a function of caffeine levels. Transfusion.60(6):1197-1211.
View at Publisher | View at Google Scholar - Tommerdahl KL, Hu EA, Selvin E, et al. (2022).Coffee Consumption May Mitigate the Risk for Acute Kidney Injury: Results From the Atherosclerosis Risk in Communities Study. Kidney international reports.7(7):1665-1672.
View at Publisher | View at Google Scholar - Caffeine. Drugs and Lactation Database (LactMed®). Bethesda (MD): National Institute of Child Health and Human Development; 2006.
View at Publisher | View at Google Scholar - Clementz GL, Dailey JW. (1988). Psychotropic effects of caffeine. American family physician. 37(5):167-172.
View at Publisher | View at Google Scholar - Górska AM, Gołembiowska K. (2015). The role of adenosine A1 and A2A receptors in the caffeine effect on MDMA-induced DA and 5-HT release in the mouse striatum. Neurotoxicity research.27(3):229-245.
View at Publisher | View at Google Scholar - Johnson DA, Ulus IH, Wurtman RJ. (1992). Caffeine potentiates the enhancement by choline of striatal acetylcholine release. Life sciences.51(20):1597-1601.
View at Publisher | View at Google Scholar - Rivera-Oliver M, Díaz-Ríos M. (2014). Using caffeine and other adenosine receptor antagonists and agonists as therapeutic tools against neurodegenerative diseases: a review. Life sciences.101(1-2):1-9.
View at Publisher | View at Google Scholar - Do HN, Akhter S, Miao Y. (2021). Pathways and Mechanism of Caffeine Binding to Human Adenosine A(2A) Receptor. Frontiers in molecular biosciences.8:673170.
View at Publisher | View at Google Scholar - Alasmari F. (20220). Caffeine induces neurobehavioral effects through modulating neurotransmitters. Saudi pharmaceutical journal : SPJ : the official publication of the Saudi Pharmaceutical Society.28(4):445-451.
View at Publisher | View at Google Scholar - McLellan TM, Caldwell JA, Lieberman HR. (2016). A review of caffeine’s effects on cognitive, physical and occupational performance. Neuroscience & Biobehavioral Reviews.71:294-312.
View at Publisher | View at Google Scholar - Lucas M, O'Reilly EJ, Pan A, et al. (2014). Coffee, caffeine, and risk of completed suicide: results from three prospective cohorts of American adults. The world journal of biological psychiatry : the official journal of the World Federation of Societies of Biological Psychiatry.15(5):377-386.
View at Publisher | View at Google Scholar - Basurto-Islas G, Blanchard J, Tung YC, et al. (2014). Therapeutic benefits of a component of coffee in a rat model of Alzheimer's disease. Neurobiology of aging. 35(12):2701-2712.
View at Publisher | View at Google Scholar - Wasim S, Kukkar V, Awad VM, Sakhamuru S, Malik BH. (2020). Neuroprotective and Neurodegenerative Aspects of Coffee and Its Active Ingredients in View of Scientific Literature. Cureus.12(8):e9578.
View at Publisher | View at Google Scholar - Fukuyama K, Kakio S, Nakazawa Y, et al. (2018). Roasted Coffee Reduces β-Amyloid Production by Increasing Proteasomal β-Secretase Degradation in Human Neuroblastoma SH-SY5Y Cells. Molecular nutrition & food research. 62(21):e1800238.
View at Publisher | View at Google Scholar - Jee HJ, Lee SG, Bormate KJ, Jung YS. (2020). Effect of Caffeine Consumption on the Risk for Neurological and Psychiatric Disorders: Sex Differences in Human. Nutrients. 12(10).
View at Publisher | View at Google Scholar - Rodak K, Kokot I, Kratz EM. (2021). Caffeine as a Factor Influencing the Functioning of the Human Body-Friend or Foe? Nutrients.13(9).
View at Publisher | View at Google Scholar - Kim B, Nam Y, Kim J, Choi H, Won C. (2012). Coffee Consumption and Stroke Risk: A Meta-analysis of Epidemiologic Studies. Korean journal of family medicine. 33(6):356-365.
View at Publisher | View at Google Scholar - Cannistraro RJ, Badi M, Eidelman BH, Dickson DW, Middlebrooks EH, et al. (2019) CNS small vessel disease: A clinical review. Neurology. 92(24):1146-1156.
View at Publisher | View at Google Scholar - Caruso P, Signori R, Moretti R. (2019). Small vessel disease to subcortical dementia: a dynamic model, which interfaces aging, cholinergic dysregulation and the neurovascular unit. Vascular health and risk management.15:259-281.
View at Publisher | View at Google Scholar - McLellan TM, Caldwell JA, Lieberman HR. (2016). A review of caffeine’s effects on cognitive, physical and occupational performance. Neurosci Biobehav Rev.71:294-312.
View at Publisher | View at Google Scholar - Rivera-Oliver M, Díaz-Ríos M. (2014). Using caffeine and other adenosine receptor antagonists and agonists as therapeutic tools against neurodegenerative diseases: a review. Life Sci.101(1-2):1-9.
View at Publisher | View at Google Scholar - Ballesteros-Yáñez I, Castillo CA, Merighi S, Gessi S. (2018). The role of adenosine receptors in psychostimulant addiction. Front Pharmacol.8(JAN):985.
View at Publisher | View at Google Scholar - Paul V and Kazi M (1992): Measuring spontaneous motor activity of rats using a vibration sensing device. Biomedicine Journal Volume 12(2).
View at Publisher | View at Google Scholar - Dunham NW and Miya TS (1957): A note on a simple apparatus for detecting neurological deficit in rats and mice. J Am pharmac Ass (Sci-Ed) 46: 208 – 209.
View at Publisher | View at Google Scholar - Popoli P, Reggio R, Pezzola A, Fuxe K and Ferre S (1998): Adenosine A1 and A2a receptor antagonists stimulate motor activity. Neurosci. Lett. 251(3): 201 – 204.
View at Publisher | View at Google Scholar - Latini S, Pazzagli M, Pepeu G and Pedata F (1996): A2 adenosine receptors: their presence and neuromodulatory role in the central nervous system. Gen. Pharmacol. 27(6): 925 – 933.
View at Publisher | View at Google Scholar - Marston HM, Finlayson K, Maemoto T, Olverman HJ, Akahane A, et al. (1998): Pharmacological characterization of simple behavioral response mediated selectively by central adenosine A1 receptors, using in vivo and in vitro techniques. J. Pharamacol. Exp. Ther. 285(3): 1023 – 1030.
View at Publisher | View at Google Scholar - Almosawi S, Baksh H, Qareeballa A, et al. (2018). Acute Administration of Caffeine: The Effect on Motor Coordination, Higher Brain Cognitive Functions, and the Social Behavior of BLC57 Mice. Behav Sci Vol 8, Page 65;8(8):65.
View at Publisher | View at Google Scholar - Muñiz JA, Prieto JP, González B, et al. C(2017). ocaine and caffeine effects on the conditioned place preference test: Concomitant changes on early genes within the mouse prefrontal cortex and nucleus accumbens. Front Behav Neurosci.11:200.
View at Publisher | View at Google Scholar - Rendón-Ochoa EA, Padilla-Orozco M, Calderon VM, et al. (2022). Dopamine D2 and Adenosine A2A Receptors Interaction on Ca2+ Current Modulation in a Rodent Model of Parkinsonism. ASN Neuro.14.
View at Publisher | View at Google Scholar - Saadawi SS, Alennabi KA, Baayo S, Fares A, Alosta N,et al. (2020). Effect of Caffeine at Different Concentrations on Behavior and Motor Activity in Mice. J Adv Med Pharm Sci. Published online. 1-11.
View at Publisher | View at Google Scholar - Fredholm BB, Svenningsson P. (2020). Why target brain adenosine receptors? A historical perspective. Park Relat Disord. 80:S3-S6.
View at Publisher | View at Google Scholar - Olopade FE, Femi-Akinlosotu OM, Adekanmbi AJ, Ighogboja OO, Shokunbi MT. (2021). Chronic Caffeine Ingestion Improves Motor Function and Increases Dendritic Length and Arborization in the Motor Cortex, Striatum, and Cerebellum. J Caffeine Adenosine Res.11(1):3-13.
View at Publisher | View at Google Scholar - Tallis J, Duncan MJ, James RS. (2015). What can isolated skeletal muscle experiments tell us about the effects of caffeine on exercise performance? Br J Pharmacol. 172(15):3703.
View at Publisher | View at Google Scholar

 Clinic
Clinic
