Case Report | DOI: https://doi.org/DOI:10.31579/2835-7957/018
Biphasic Synovial Sarcoma of the Neck – A Rare Case Report with Review of Literature
1 The Division of Pediatric Otorhinolaryngology-Head and Neck Surgery, The Cincinnati Children’s Medical Hospital, Ohio, USA.
2 Doctoral Degree Program in Neuroscience, Yeditepe University, Istanbul, Turkey.
3 Medicine Program, Ege University Faculty of Medicine, Izmir, Turkey.
4 Department of Otorhinolaryngology, Head and Neck Surgery, Private Derindere Hospital, Istanbul, Turkey.
5 Department of Otolaryngology, Head and Neck Surgery, Izmir Tepecik Training and Research Hospital, Izmir, Turkey.
*Corresponding Author: Aynur Aliyeva, Doctoral Degree Program in Neuroscience, Yeditepe University, Istanbul, Turkey.
Citation: Aliyeva A., Karimov Z Fidan R. Alekberli, Yagiz O., (2023), Biphasic Synovial Sarcoma of the Neck – A Rare Case Report with Review of Literature, Clinical Reviews and Case Reports, 2(2); DOI:10.31579/2835-7957/018
Copyright: © 2023, Aynur Aliyeva. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: 15 March 2023 | Accepted: 05 April 2023 | Published: 11 April 2023
Keywords: biphasic sarcoma; head and neck tumors; synovial sarcoma
Abstract
Synovial Sarcomas are malignant tumors of mesenchymal origin. It is rarely seen in the head and neck region. There is male dominance among the patients with SS. In our case, 51 years old female patient was admitted to the ENT clinic with the complaint of painless swelling on the right neck that had progressively enlarged in the last two months. After the examinations, a diameter of 7x5.5 cm of mass was observed in the posterior sternocleidomastoid muscle in the MRI. After the non-diagnostic FNA and pan-endoscopy, the tumor was surgically removed. The histopathology diagnosis of the neck mass was Biphasic Synovial Sarcoma. Chemoradiotherapy was recommended after the surgery.
No local recurrence or distant metastasis was found in the postoperative 4nd-year control assessment. This study emphasizes the importance of a multidisciplinary approach and raises awareness in diagnosing and treating a very rare case of Biphasic Synovial Sarcoma in the head and neck region.
Introduction
Synovial Sarcomas (SS) are high-grade malign soft tissue sarcomas derived from mesenchymal pluripotent cells and commonly located in the deep soft tissues of the lower extremities [1,2]. SS is rarely seen in the head and neck region with 1.9-3.5% [3–5]. Although it morphologically resembles synovial tissue but is not originate from synovium [2]. There are two histological subtypes of the SS, including epithelial and spindle cell/sarcomatous components: monophasic and biphasic. Monophasic SS is more common than biphasic [2,6]. Treatment of SS is surgical excision of the lesion and radiochemotherapy [2,7]. Since primary biphasic SS in the neck region is extremely rare, we think our case will contribute to the literature.
Case Presentation
A 51-year-old woman was admitted to the ENT and HNS clinic with the complaint of painless swelling on her right neck that had progressively enlarged in the last two months. In neck ultrasonography (USG) examinations, a hypo-isoechoic solid mass in the posterior of the right sternocleidomastoid (SCM) muscle was determined, containing areas of cystic degeneration in some places, compared to the surrounding tissue. The mass measured 7.5x5.5 cm, starting from the area matching the parotid gland localization on the right and continuing between the posterior spinal muscles in the contrast-enhanced neck magnetic resonance image (MRI). It shows diffusion restriction in the T2 series and a distinct heterogeneous contrasting pattern in postcontrast examination. (Figures 1.).
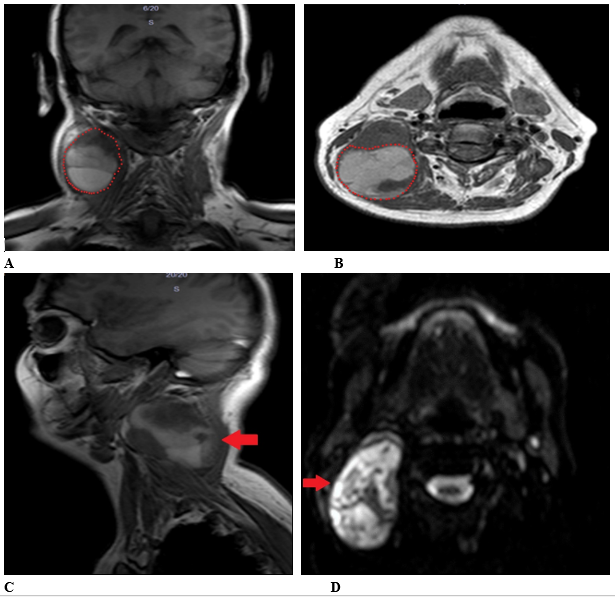
Figure 1: Magnetic resonance image (MR) of the neck. Neck mass invading neighboring muscle.
(A) T1 - Coronal axis. Red dots show the tumor border.
(B) T1 - Axial axis. Red dots show the tumor border.
(C) T1 - Sagital axis. Red arrow show the tumor localization.
(D)T2 – Axial axis. Red arrow show the tumor localization.
Tuberculosis and lymphadenitis were considered primary differential diagnoses. The second possible diagnoses were lymph node with necrotic component and infectious mononucleosis. Then, pan endoscopy and fine needle aspiration biopsy (FNAB) was performed. Panendoscopy was interpreted as usual. Upon the suspicious cytology result of FNAB, total excision of the mass from the right neck was planned for the patient.
Orotracheal intubation was performed under general anesthesia. The mass was approximately 8x8 cm in diameter and placed behind the SCM muscle on the right side of the neck. A 10 cm horizontal incision was made over the mass. The skin, subcutaneous tissue, and platysma were passed, and the lesion was reached. The mass was dissected from the surrounding tissues. The lesion is located on the paravertebral fascia at the base. There was a pedicle-like tract extending to the jugular foramen, skull base, from under the SCM muscle; as a result of this, it was cut and ligated. The largest dimension is 5 cm, in which tumor integrity was still observed. The mass drained during excision, and bleeding occurred, but it was under control. The lesion was excised totally, and hemostasis was obtained. A hemovac drain was placed. The layers were closed following the anatomy. No surgical complications and hemostatic instabilities were observed postoperatively.
Afterward, the pathological result of the mass was described as Biphasic Synovial Sarcoma. There were epithelial and mesenchymal developmental patterns. In the epithelial areas (“+”, positive; “-”, negative): pankeratin (+), high-molecular-weight kininogen (HMWK) (+), Bcl-2 (+), CD99 (-), S-100 (-), desmin (-), mammary serum antigen (MSA) (-), CD34 (-), thyroid transcription factor-1 (TTF1) (-), calcitonin (-), and amyloid (-). The Ki67 staining rate was 30%. Morphological and immunohistochemical findings suggest Synovial Sarcoma (SS) for diagnosis.
Furthermore, it was advised to investigate specific translocation for SS to a definitive diagnosis. Then chemoradiotherapy treatment was recommended to the patient according to the decision of the oncology council. After four years of multimodal therapy, the patient does not exhibit signs of local recurrence and distant metastasis.
Discussion
Synovial Sarcoma (SS) is an uncommon soft tissue malignancy accounting for 8–10% of soft tissue neoplasms that are more frequent in the younger population (15–37 years old), and the male/female ratio is 3:2 1,3,6–10. Our case is 50 years old woman, making this report valuable for literature.
Translocation between chromosomes 18 (SYT gene) and X (SSX1 or SSX2 genes) and t(X;18) (p11.2; q11.2) are associated with SS [2,11,12]. S-100 was staining poorly in immunohistochemical studies, which is similar to our case; however, other sating results vary from case to case [3,13].
There are two histomorphological subtypes of SS: monophasic and biphasic. The first show only epithelial features; the second has both sarcomatous/spindle cells and epithelial components [1,8]. There is no significant difference in survival outcome between the two subgroups [6,14].
Patients with SS of the head and neck usually present with painless mass and slow progressive growth; hence it has a vague clinical presentation [6,8,15]. Also, otalgia, hoarseness, dysphagia, odynophagia, foreign body sensation, or bleeding may be present depending on tumor localization [6,15]. Head and neck SSs can place in the submandibular, parapharyngeal, supraclavicular, hypopharyngeal, para-SCM, skull base, sinonasal tract, scalp, parotid gland, paraspinal, paravertebral, thyroid gland, and gingiva areas [8,9,16–20]. Harb et al. reported no association between localization and recurrence, but mortality was significantly higher in the paraspinal, skull base, and sinonasal SS [6]. The recurrence rate varies from 5.5% to 45.5% [2–4]. The recurrence was related to overall survival (OS) [21].
The mean tumor size is 0.5-7.0 cm [3,4,10]. Studies found that tumors with >5cm diameter are a risk factor for distant metastasis, local recurrence, and OS [7,10]. On the other hand, Gopalakrishnan et al. reported no significant differences in progression-free survival and OS while considering >5cm tumor diameter as a cut-off value [2]. Macroscopic examination after the excision showed that our case’s tumor diameter was 5,5 cm. Studies showed that wide surgical excision and negative surgical margin are prognostic factors for recurrence and survival [8,22,23].
Carillo et al. reported that microcalcification might be seen in imaging and histologic exam of ~33% of SS 1. However, it was not found any calcification in our case.
SS generally metastasize hematogenously; however, studies reported that tumor could spread to lymph nodes in 15-20% of cases [10,24,25]. But, lymph node involvement was unrelated to prognosis and local recurrence [14]. Therefore, no evidence exists for prophylactic neck dissection to improve outcome [14]. Crowson et al. reported that distant metastasis is associated with poor survival [14]. Additionally, OS is similar between male and female patients [6]. Although SS grows slowly, it has a poor prognosis with an average survival rate at five years of 25–66% and a 10-year survival rate of 11–53% [1,10]. In our case, there is no evidence of tumor recurrence and metastasis for four years. Multimodal treatment is the main option for the patient with biphasic SS [2,5,16,26].
This study emphasizes the importance of a multidisciplinary approach and raises awareness in diagnosing and treating a very rare case of Biphasic Synovial Sarcoma in the head and neck region.
Consent for publication:
The patient consented to the publication of this case report.
Availability of data and material: Data sharing does not apply to this article as no datasets were generated or analyzed during the current study.
Competing interests:
The authors declare no conflict of interest.
Funding:
The authors have no financial relationship to declare.
Authors' contributions:
A.A.: Study design, examination of patients, data collection and analysis, manuscript development, and review of the final manuscript, supervision.
Z.K: Study design, manuscript development, and review of the final manuscript.
F.R.A: Study design, manuscript development, and review of the final manuscript.
O.Y: Study design, manuscript development, and review of the final manuscript.
References
- Carrillo R, Rodriguez-Peralto JL, Batsakis JG. (1992).Synovial sarcomas of the head and neck. Ann Otol Rhinol Laryngol.101(4):367-370.
View at Publisher | View at Google Scholar - Gopalakrishnan V, Amini B, Wagner MJ, et al. (2017). Synovial Sarcoma of the Head and Neck: A Single Institution Review. Sarcoma.
View at Publisher | View at Google Scholar - Al-Daraji W, Lasota J, Foss R, Miettinen M. (2009).Synovial sarcoma involving the head: analysis of 36 cases with predilection to the parotid and temporal regions. Am J Surg Pathol.33(10):1494-1503.
View at Publisher | View at Google Scholar - Stanbouly D, Litman E, Lee KC, Philipone E. (2021).Synovial sarcoma of the head & neck: A review of reported cases in the literature. J Stomatol Oral Maxillofac Surg. 122(5):505-510.
View at Publisher | View at Google Scholar - Mullen JR, Zagars GK. (1994). Synovial sarcoma outcome following conservation surgery and radiotherapy. Radiother Oncol. 1994;33(1):23-30.
View at Publisher | View at Google Scholar - Harb WJ, Luna MA, Patel SR, Ballo MT, Roberts DB. (2007).Survival in patients with synovial sarcoma of the head and neck: Association with tumor location, size, and extension. Head Neck. 29(8):731-740.
View at Publisher | View at Google Scholar - Wushou A, Miao XC. (2015). Tumor size predicts prognosis of head and neck synovial cell sarcoma. Oncol Lett. 9(1):381.
View at Publisher | View at Google Scholar - Sturgis EM, Potter BO. (2003). Sarcomas of the head and neck region. Curr Opin Oncol. 15(3):239-252.
View at Publisher | View at Google Scholar - Doval DC, Kannan V, Mukherjee G, Shenoy AM, Shariff MH, et al. (1997),Synovial sarcoma of the neck. Eur Arch Otorhinolaryngol. 254(5):246-250.
View at Publisher | View at Google Scholar - Mallen-St Clair J, Arshi A, Abemayor E, St John M. (2016). Synovial cell sarcoma of the head and neck: an analysis of 167 cases using the SEER database. JAMA Otolaryngol Head Neck Surg. 142(6):576.
View at Publisher | View at Google Scholar - Kawai A, Woodruff J, Healey JH, Brennan MF, Antonescu CR, et al. (1998).SYT-SSX gene fusion as a determinant of morphology and prognosis in synovial sarcoma. N Engl J Med. 1998;338(3):153-160.
View at Publisher | View at Google Scholar - Clark J, Rocques PJ, Crew AJ, et al. (1994).Identification of novel genes, SYT and SSX, involved in the t(X;18)(p11.2;q11.2) translocation found in human synovial sarcoma. Nat Genet. 7(4):502-508.
View at Publisher | View at Google Scholar - Khademi B, Daneshbod Y, Negahban S, et al. (2006).Biphasic parapharyngeal synovial sarcoma: a cytologic and immunocytologic report of a case. Cytojournal. 2006;3.
View at Publisher | View at Google Scholar - Crowson MG, Lalich I, Keeney MG, Garcia JJ, Price DL. (2015). Clinicopathologic factors and adjuvant treatment effects on survival in adult head and neck synovial cell sarcoma. Head Neck.37(3):375-380.
View at Publisher | View at Google Scholar - Bukachevsky RP, Pincus RL, Shechtman FG, Sarti E, Chodosh P. (1992).Synovial sarcoma of the head and neck. Head Neck. 14(1):44-48.
View at Publisher | View at Google Scholar - Fonseca AS, Azevedo ACA, Magalhães FM, De Andrade NA. (2014).Synovial Sarcoma in Head and Neck: A Case Report. Int Arch Otorhinolaryngol. 18(1):87.
View at Publisher | View at Google Scholar - Rao GV, Sravya T, Sivaranjani Y, Bhat VR. (2014).Primary biphasic synovial sarcoma of gingiva: Report of a rare case. J Oral Maxillofac Pathol. 18(1):77-79.
View at Publisher | View at Google Scholar - Wadhwan V, Malik S, Bhola N, Chaudhary M. (2011).Biphasic synovial sarcoma in mandibular region. J Oral Maxillofac Pathol.15(2):239-243.
View at Publisher | View at Google Scholar - Foreman SM, Stahl MJ. (2011).Biphasic synovial sarcoma in the cervical spine: Case report. Chiropr Man Therap. 19(1).
View at Publisher | View at Google Scholar - Jang KS, Min KW, Jang SH, et al. (2007).Primary synovial sarcoma of the thyroid gland. J Korean Med Sci. 22 Suppl(Suppl).
View at Publisher | View at Google Scholar - Owosho AA, Estilo CL, Rosen EB, Yom SHK, Huryn JM,et al. (2017).A clinicopathologic study on SS18 fusion positive head and neck synovial sarcomas. Oral Oncol. 66:46.
View at Publisher | View at Google Scholar - Kraus DH, Dubner S, Harrison LB, et al. Prognostic Factors for Recurrence and Survival in Head and Neck Soft Tissue Sarcomas.
View at Publisher | View at Google Scholar - Vay J, O’sullivan B, Catton C, et al. (1994). An Assessment of Prognostic Factors in Soft-Tissue Sarcoma of the Head and Neck. Arch Otolaryngol Neck Surg. 120(9):981-986.
View at Publisher | View at Google Scholar - Moore DM, Berke GS. (1987).Synovial sarcoma of the head and neck. Arch Otolaryngol Head Neck Surg. 113(3):311-313.
View at Publisher | View at Google Scholar - Roth JA, Enzinger FM, Tannenbaum M. SYNOVIAL SARCOMA OF THE NECK: A FOLLOWUP STUDY OF 24 CASES.
View at Publisher | View at Google Scholar - Bergh P, Meis-Kindblom JM, Gherlinzoni F, et al. (1999). Synovial sarcoma: Identification of low and high risk groups. Cancer. 85(12):2596-2607.
View at Publisher | View at Google Scholar

 Clinic
Clinic
